CHEM1831 Lecture Notes - Lecture 15: Tennis Ball, Heat Capacity, Thermodynamics
Document Summary
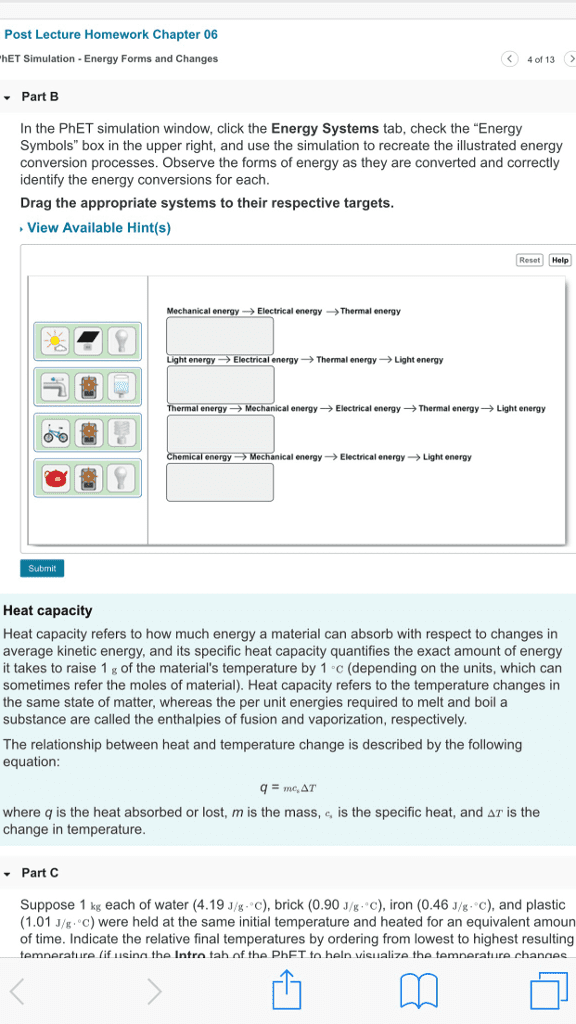
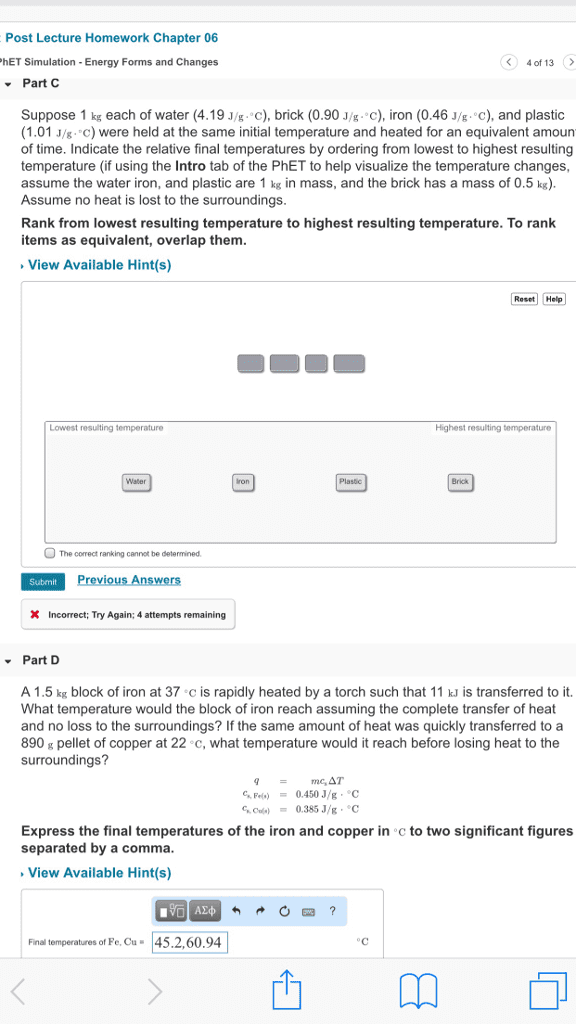
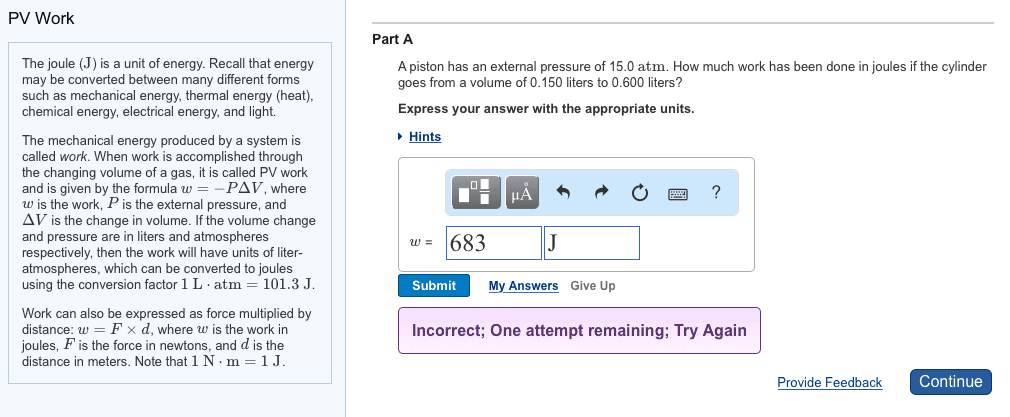
Is the study of the transformations of heat to and from other forms of energy: the laws of thermodynamics and their consequences apply to all chemical and physical processes. The other forms of energy can be mechanical energy, electrical energy and the energy stored in chemical vbonds, hence thermodynamics is especially relevant to chemistry and biology. Thermodynamics concerns itself with the system we are studying, everything else is called the surroundings. In biological or biochemical research the system might be a whole organism, a cell or part of a cell depending on the detais of the research or problem you are studyinh. We specifify a process that affects the system and we observe or calculate energy changes associated withh that process. When you specify a process you need to specify all the conditions that can affect the energy change in the process.