CHEM1100 Lecture Notes - Lecture 9: Intermolecular Force, Horse Length, Partial Pressure
45 views4 pages
Document Summary
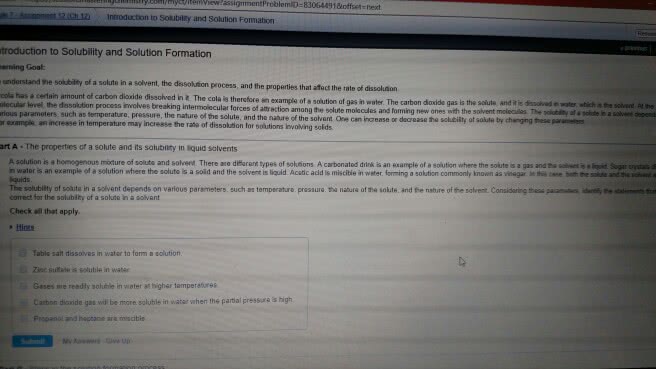
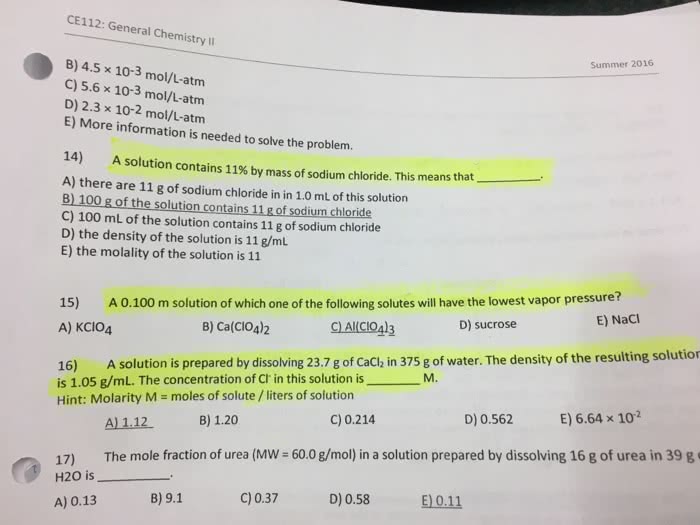
Is the maximum amount of solute (minor component) that will dissolve in a given amount of solvent (major component) at a specific temperature. When mentos is added to coke- quick release of gas. When you open the coke, it is not in equilibrium, but the mentos drives the equation to the left. Heating drives co2 off, leading to more carbonate which reacts with dissolved ca2+ to form. Note: henry"s law is not obeyed when a reaction occurs (1m3 = 103l) 2- (aq) + co2 (aq) + h2o (l) 2hco3. Liquid-liquid solutions: solute-solvent molecule interactions must be stronger than solute-solute molecule. Balance of interactions ultimately dictates solubility (-oh) functional group dominates the properties of the molecules of methanol, ethanol and propanol. Molecule contains both polar and non-polar groups- balance. O and n overrides the polarity of the methyl functional groups. It can be prepared by dissolving a solid at high temperatures (more substance dissolved) and then slowly cooling this solution.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232