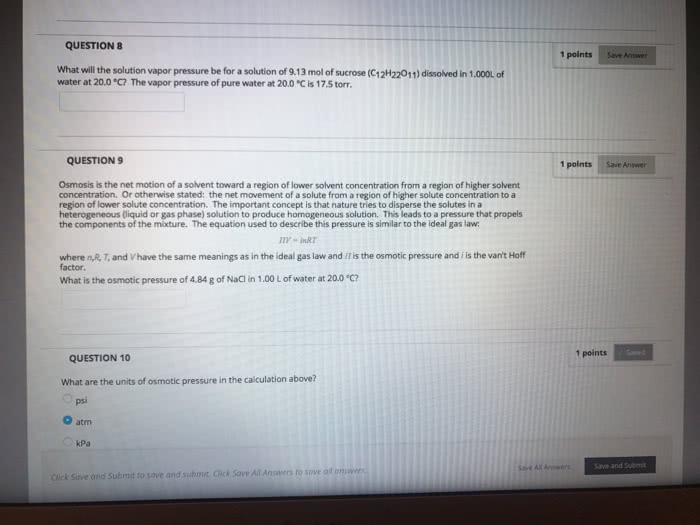
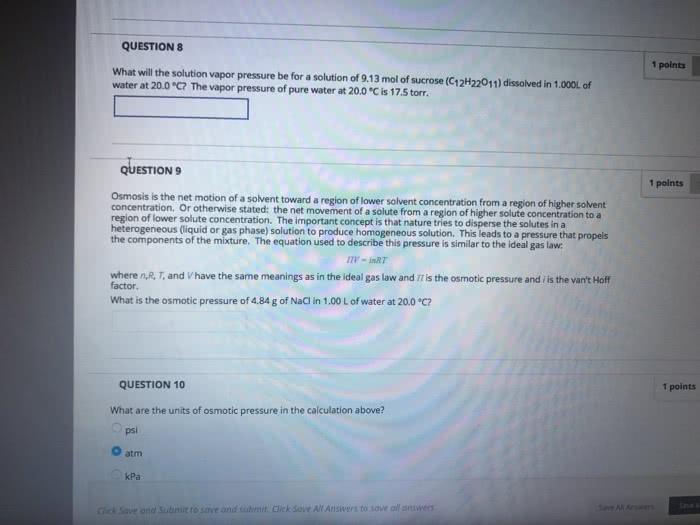
CHEM101 Lecture Notes - Lecture 12: Hemolysis, Gas Constant, Osmosis
Document Summary
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
Below, is a completed lab. After completing this lab, I was asked the following question:
What changes would you make, if any, if you did this lab again?
I'm having a hard time thinking of what I would do different. Please help!
Observations & Questions for Part 1
Record your observations and your time and temperature data in tables. Use one table for the pure water and one table for the salt solution.
| Data Table 1: Pure Water and Salt Solution | ||||
| Seconds | Distilled H20 Room temp | Distilled H20 Ice bath | Saltwater Room temp | Saltwater Ice bath |
| 0 | 23 | 23 | 17 | 23 |
| 30 | 23 | 10 | 20 | 7 |
| 60 | 23 | 4 | 22 | 1 |
| 90 | 22.5 | 1 | 22 | 0 |
| 120 | 22.5 | 0 | 22 | 0 |
| 150 | 0 | 22 | -1 | |
| 180 | -1 | 22 | -1 | |
| 210 | -1 | 22 | -2 | |
| 240 | -2 | -2 | ||
| 270 | -1 | -2 | ||
| 300 | -1 | -2 | ||
| 330 | -1 | -2 | ||
| 360 | -1 | |||
| 390 | 0 | |||
| 420 | 0 | |||
| 450 | 0 | |||
| 480 | 0 | |||
| 510 | 0 | |||
| 540 | ||||
| 570 | ||||
| 600 | ||||
| 630 | ||||
| 660 | ||||
Make two graphs of your data. On one graph plot the data from the pure water. On the other graph plot the data from the salt solution. On both plot temperature on the y-axis and time on the x-axis.
A. Record the freezing point of the pure water and the freezing point of the salt solution.
Freezing point of salt Sol: -2 degree Celsius
Freezing point of pure H2O: 0 degree Celsius
B. How do these two freezing points compare?
The freezing point of salt water is lower than the freezing
point of fresh water. Salt in the water lowers the freezing point
of water.
C. What are some practical applications of freezing point depression, boiling point elevation, and vapor pressure lowering?
Some practical applications of freezing point depression
are antifreeze in a radiator and salt on the road used to melt
ice in the winter. Some practical applications of boiling point
elevation are a sealed container and possibly a pressure cooker.
Some practical applications of vapor pressure lowering are freeze
drying and steam engines.
Questions - Part 2
To what biological structure is the dialysis bag comparable? How is it similar? How is it different?
The plasma membrane is comparable to the dialysis bag.
The similarity between them is that they are both semipermeable.
The difference is the dialysis bag acts as a permeable membrane,
however, purely based on its pore size, the small molecules are
able to get through but the bigger molecules are unable to. The
plasma membrane is a more complex system that uses both active
and passive transport to allow the molecule to move through.
In biological systems if a cell is placed into a salt solution in which the salt concentration in the solution is lower than in the cell, the solution is said to be hypotonic. Water will move from the solution into the cell, causing lysis of the cell. In other words, the cell will expand to the point where it bursts. On the other hand, if a cell is placed into a salt solution in which the salt concentration in the solution is higher than in the cell, the solution is said to be hypertonic. In this case, water will move from the cell into the solution, causing cellular death through crenation or cellular shrinkage. In your experiment is the Karo