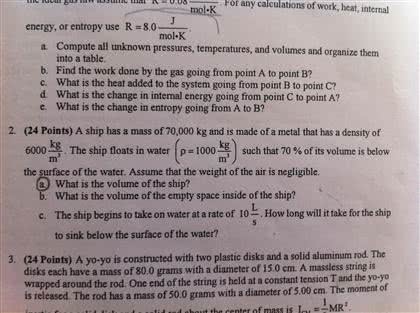PHYS1001 Lecture Notes - Lecture 15: Boltzmann Constant, Carnot Cycle, Chemical Energy
Document Summary
The total entropy of an isolated system can never decrease over time. It is impossible for any process to have as its sole result the transfer of heat from a cooler to a hotter body. For an isolated system, the direction of spontaneous change is from: A situation of less randomness to greater randomness. This refers to the notion of entropy. The units of entropy are j/k dv = dq. Macroscopically, s = q for reversible processes. The change of entropy is path independent. Natural thermodynamic processes occur in only one direction. Reversible processes do exist, however, they only occur in systems which are very close to equilibrium with their surroundings. Heat engines use thermodynamic properties to convert chemical potential energy / heat energy (depending on how you look at it) into mechanical work. Sometimes we are more interested in the thermal activity of an engine than the finer workings of the mechanics.