CHEM 1F92 Lecture Notes - Lecture 12: Titration Curve, Equivalence Point, Methyl Red
Document Summary
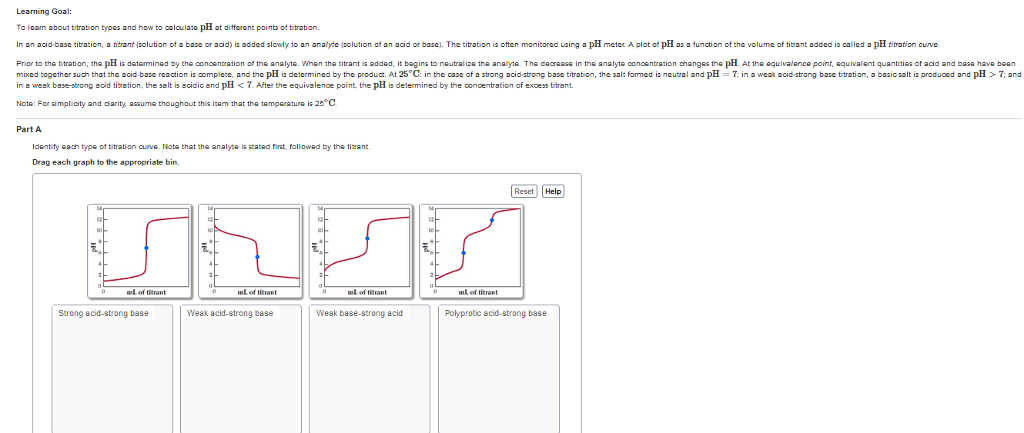
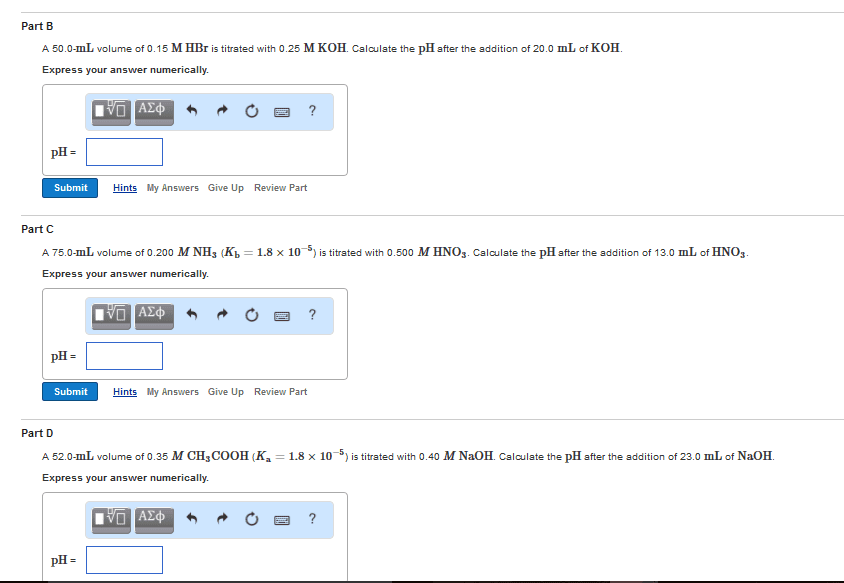
Bronsted acid donates a proton (to water). Bronsted base accepts a proton (from water). Hint: ph=7 at equivalence point for strong acid/ strong base titration only! Equivalence point: point at which the concentration of protons = concentration of hydroxide ions. Ph is determined by the concentration of excess unreacted [h+] or [oh-] Ph<7 at equivalence point for strong acid/ weak base titration. Ph>7 at equivalence point for strong base/ weak acid titration. Phenolphthalein is used to determine the equivalence point of ph>7. Methyl red is used to determine the equivalence point of ph<7. Endpoint: colour change tells us to stop titration. Endpoint should be close to the equivalence point. Before equivalence point, ph<7, and we have [h3o+] in excess.