CHEM 1F92 Lecture Notes - Lecture 8: Acid Hydrolysis, Hydrolysis, Equilibrium Constant
Document Summary
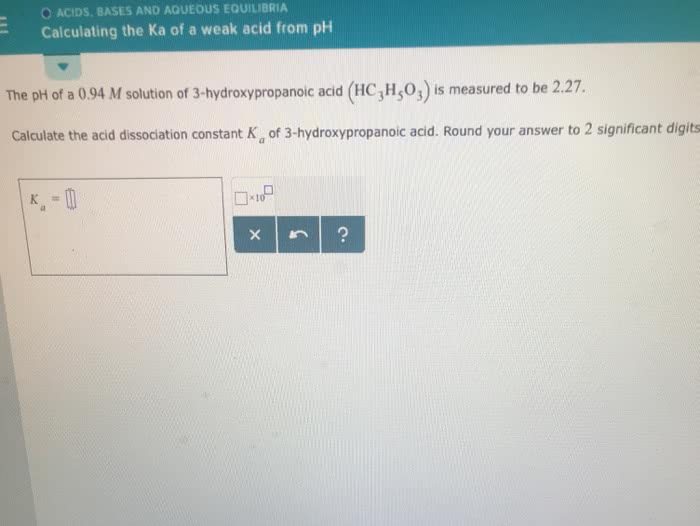
Weak acids and bases dissociate only partially in aqueous solutions. Stronger acids (cid:3247)like(cid:3248) to react with stronger bases, to produce weaker bases and weaker acids. Equilibrium constant, k, for acid dissociation is ka. The weaker the acid, the smaller the acid ionization constant, ka. H2o + hf f- + h3o+ If hf does not dissociate by much, then x<< 0. 50 m and (0. 50 m - x) = ~ 0. 50 m. Dilution means to reduce the concentration of a solute by adding solvent. Le chatelier"s principle: the equilibrium will shift towards the product side if an applied stress reduces [products] Dilution of a weak acid will shift equilibrium. Strong acids and bases are 100% ionized. The number of decimal places of a ph is equal to the number significant figures of the corresponding concentration. At very low concentrations, weak acids and bases ionize completely. Acid hydrolysis: ha + h2o a- + h3o+