CHMI-1041EL Lecture Notes - Lecture 4: Sodium Hydroxide, Transition Metal, Ion
Document Summary
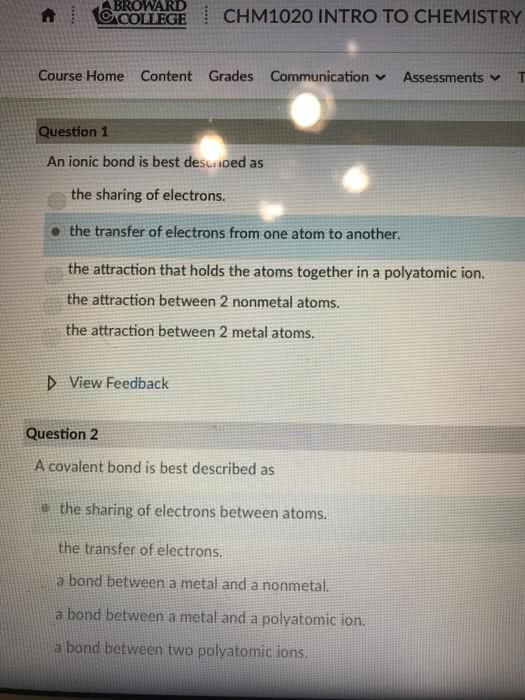
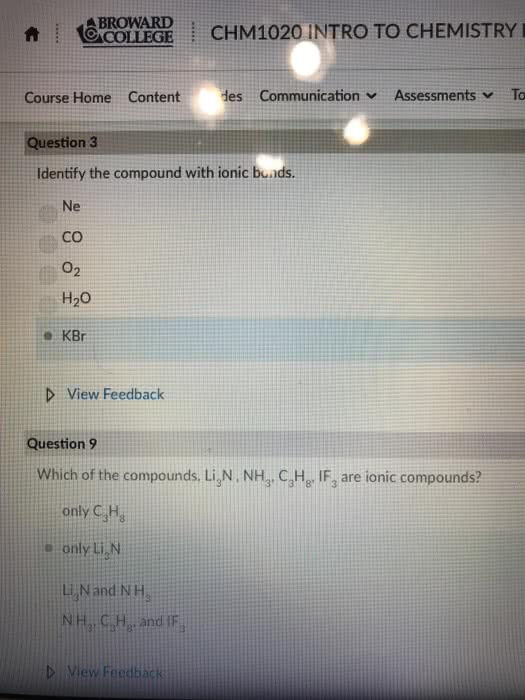
A chemical bond is an attractive force that holds two atoms together in a more complex unit. Form as a result of interactions between electrons found in the combining atoms. Ionic bonds: formed through the transfer of one or more electrons from one atom or group of atoms to another. An ionic compound is a compound in which ionic bonds are present: covalent (molecular) bond: formed through the sharing of one or more electrons between two atoms. Molecular compound (covalent) is a compound where two atoms are joined though covalent bonds. Most bonds are not completely ionic or covalent, they have some degree of both characteristics. Two fundamental concepts of chemical bonding: not all electrons in an atom participate in bonding. Those that participate are called valence electrons: certain arrangements of electrons are more stable than others, as is explained by the octet rule.