CHEM 110 Lecture Notes - Lecture 5: Alkene, Alkyne, Stereoisomerism

Organic Structures
CHEM 110: ORGANIC CHEMISTRY
THE OTHER HYDROCARBONS!!!!!!!!!
Cycloalkenes: “unsaturated”, sigma and pi bonds (CnH2n-2)
CH2=CH2
CH2=CHCH3 INCREASING VISCOSITY ↓
CH2=CHCH2CH3
CH2=CH(CH2)2CH3
*DOUBLE BONDS MUST INCLUDE CARBON 1 AND CARBON 2, THEY TAKE
PRIORITY*
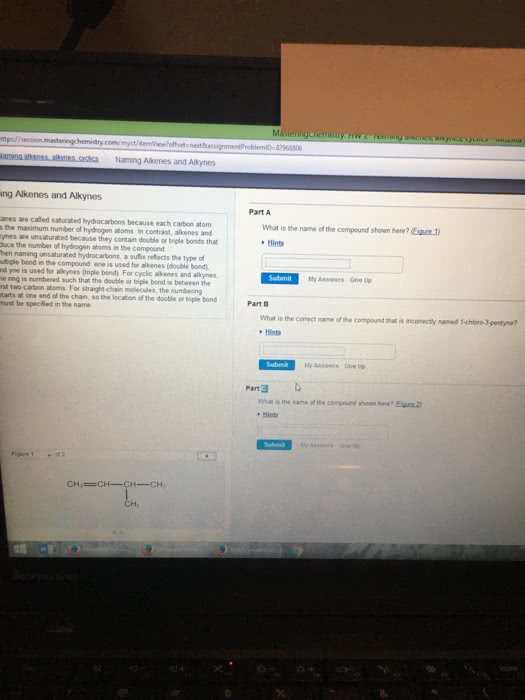
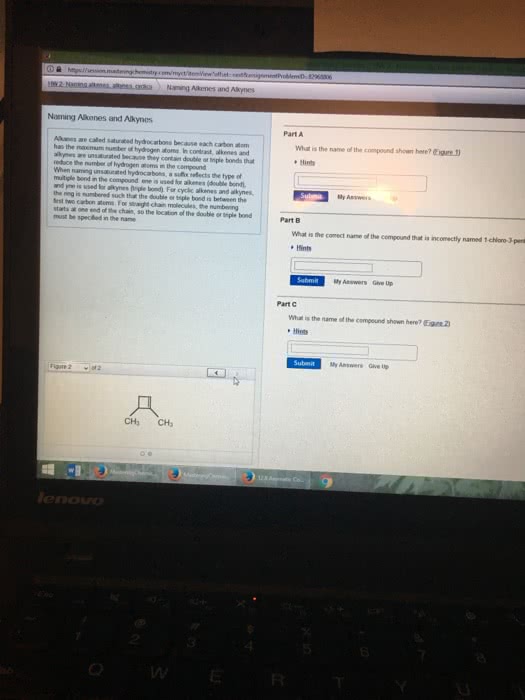
Naming Alkenes:
5-ethyl-2-methyl-2-octene
put the double bonded carbon at the end in front of the name*
3-ethylcyclopentene
3,6-dimethyl-1,5-heptdiene
find more resources at oneclass.com
find more resources at oneclass.com

37
CHEM 110 Full Course Notes
Verified Note
37 documents
Document Summary
*double bonds must include carbon 1 and carbon 2, they take. 5-ethyl-2-methyl-2-octene put the double bonded carbon at the end in front of the name* Alkene: fixed, rigid double bonded system, it is difficult to rotate. Cis vs trans: same base name, differentiating feature with their structures. Axis of the double bond: if the two carbon groups are on the same side of the axis, it is cis, if it is on the opposite sides, it is trans. (draw the line through the double bond) Alkynes: unsaturated , sigma and 2 pi bonds (cnh2n-2) Naming: same idea as alkenes, lowest # for our alkyne, the triple bond takes priority. Benzenes: unsaturated , sigma and 3 pi bonds (c6h6) e. g. methyl group: methylbenzene. Benzene: bonding orbitals filled, no antibonding orbitals = super stable. Criteria for aromaticity: monocyclic (1 ring, planar (all atoms sp2) flat, 4n+2 pi electrons (n > 0) Different compounds with the same molecular formula.