CHEM 120 Lecture 8: Thermodynamics+kinetics

26
CHEM 120 Full Course Notes
Verified Note
26 documents
Document Summary
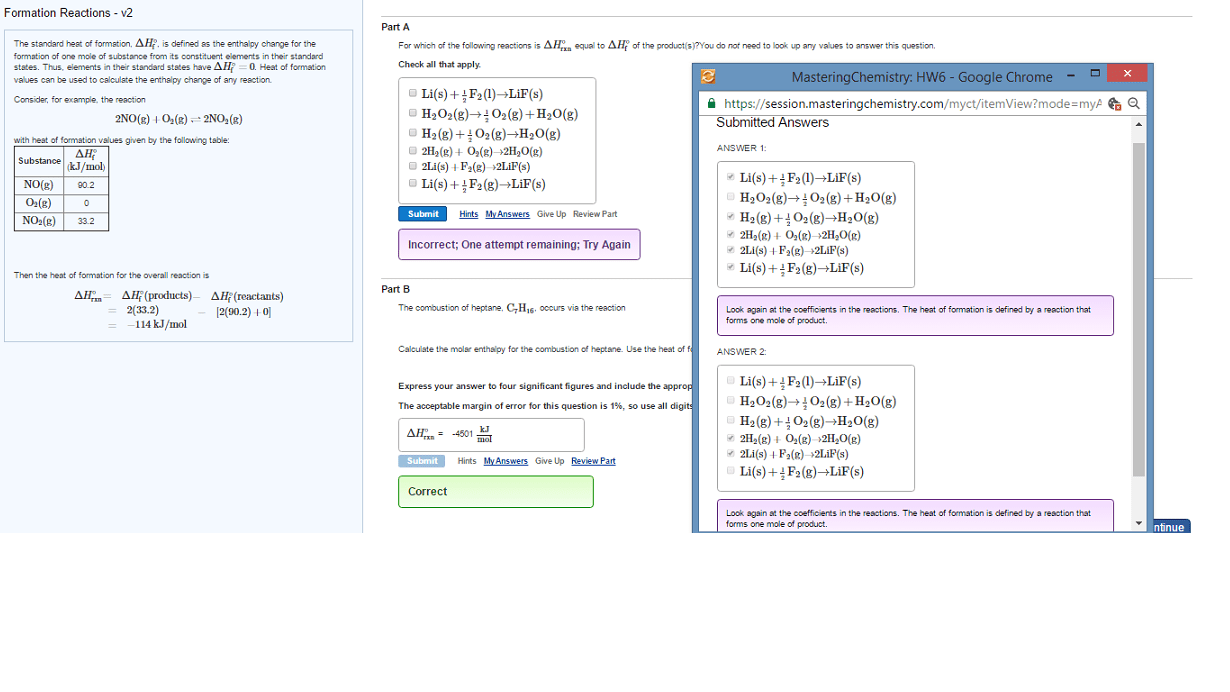
Enthalpy is an extensive property (related to the amount of substance) N2(g)+ o2(g) 2 no(g) h = +180. 50 kj. Using the above information, determine the h for the following reaction. H total= h1 + h2+ h3 + hn. Step by step reactions to determine total h. For a gas, p= 1 bar, ideal behaviour. The enthalpy change that occurs in the formation of 1 mole of a substance in its standard state, from the elements it is made of in their standard states. Enthalpy of formation is represented by fh . Na (s) + cl2 (g) nacl (s) Fh =0 for a pure element in its standard state. Since enthalpy is extensive, we need to multiply fh by the number of moles of a substance. Chemical kinetics measures the rate of reactions in relation to the concentration of substances changing over time. H2 (g)+ f2 (g) 2hf (g)