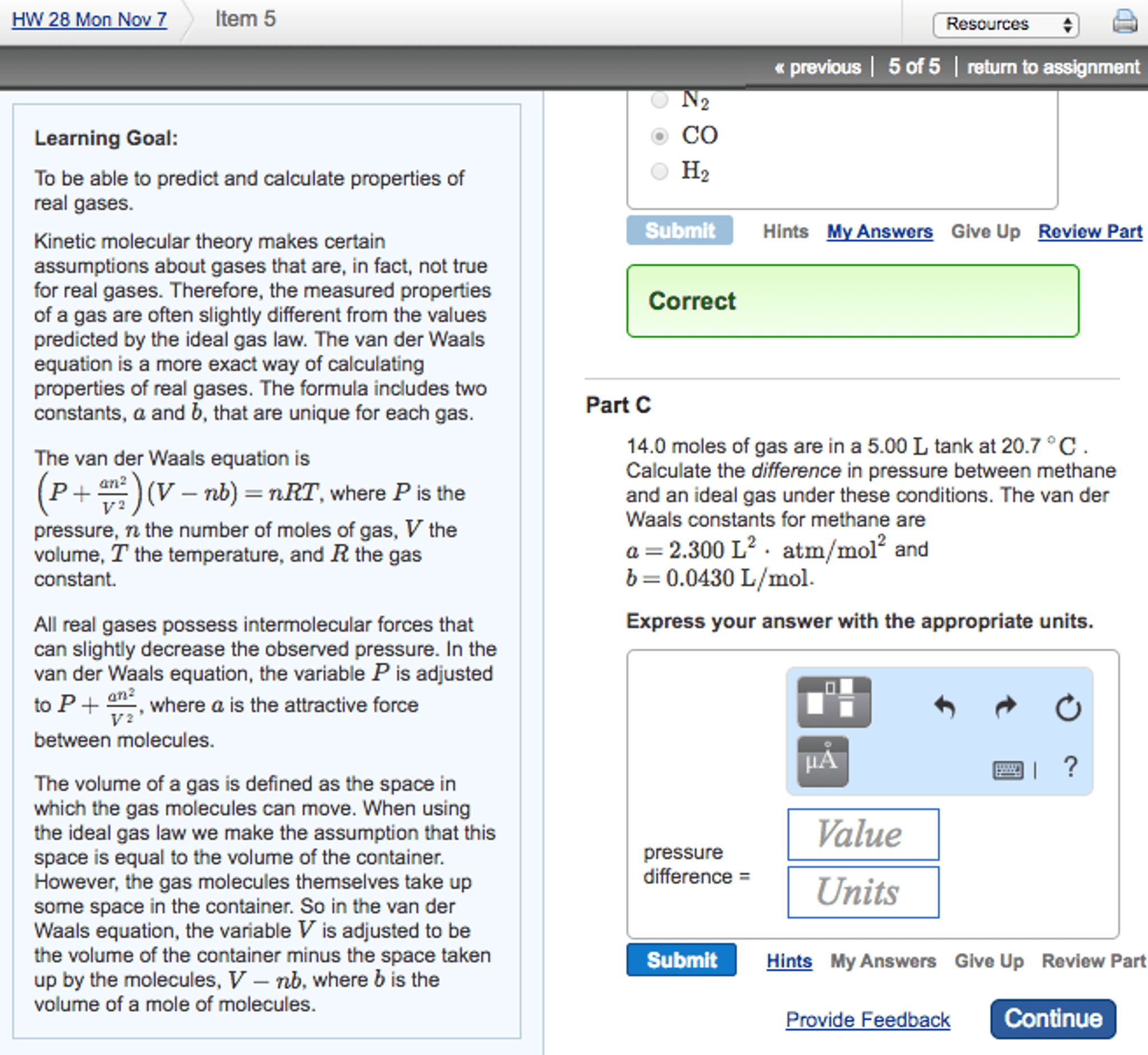
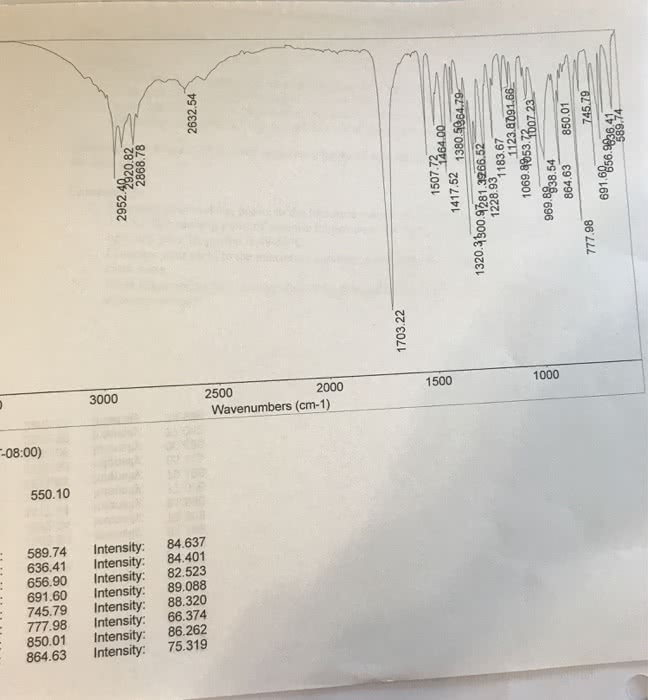
I need a table for the significant peaks of the final product.

xperiment 10: Resolution of Racemic Ibuprofen and (adapted from McCullagh, Jornal of Chemical Ecarion. Determination of Optical Purit Question to be addressed by doing this experiment: H Vol 85, Jully 2008, P 419) another? How do we separate one enantiomer from Required Reading: Mohrig, Enantiomeric Analysg. Chapter 17, Sections 1-3 (pp. 240-248), "Optical Activity and 17 Before you come to lab: Prepare your prelab with introduction, safety data for the reagents to be used, answers to pre-lab questions, and procedure. BACKGROUND INFORMATION The ability to produce enantiomerically pure compounds is crucial to the fine chemicals ind ustries (pharmaceuticals, agrochemicals, fragrances, etc). This ability is probably most mportant to the pharmaceutical industry, where approximately 80% of the drugs under development are chiral. Enantiomerically pure compounds can be produced by one of two methods- resolution and asymmetric synthesis. Resolution is the process in which the two enantiomers of a racemic mixture are separated. Asymmetric synthesis involves the use of a Chiral reagent or catalyst to create a new optically active molecule. In this experiment, you will separate a racemic mixture of ibuprofen into its R and S enantiomers. Ibuprofen belongs to a class of pharmaceuticals called non-steroidal anti- inflammatories, or NSAIDs. It is the active ingredient in Motrin and Advil, along with their generic equivalents. It is primarily used for pain relief and fever reduction. Of the two enantiomers, only one, the (S)-C+)-enantiomer, has the desired pharmacological activity, while the R enantiomer is inactive. Since the R-enantiomer is not harmful, most commercial ibuprofen is sold as a racemic mixture, but recent research indicates that using only the S enantiomer might be beneficial, so several companies are developing methods to produce the enantiomerically pure form of ibuprofen and related compounds. The resolution you will be conducting works by temporarily converting the pair of enantiomers, which have identical physical properties, into a pair of diastereomers, with different physical properties, by using a single enantiomer of a second chiral compound (the resolving agent). Ibuprofen is a carboxylic acid, which will react with an amine to make an ammonium carboxylate salt: NH (S,S) salt, insoluble in aqueous solution racemic ibuprofen (S)H-+a-phenylethylamine (R,S) salt, soluble in aqueous solution xperiment 10- Resolution