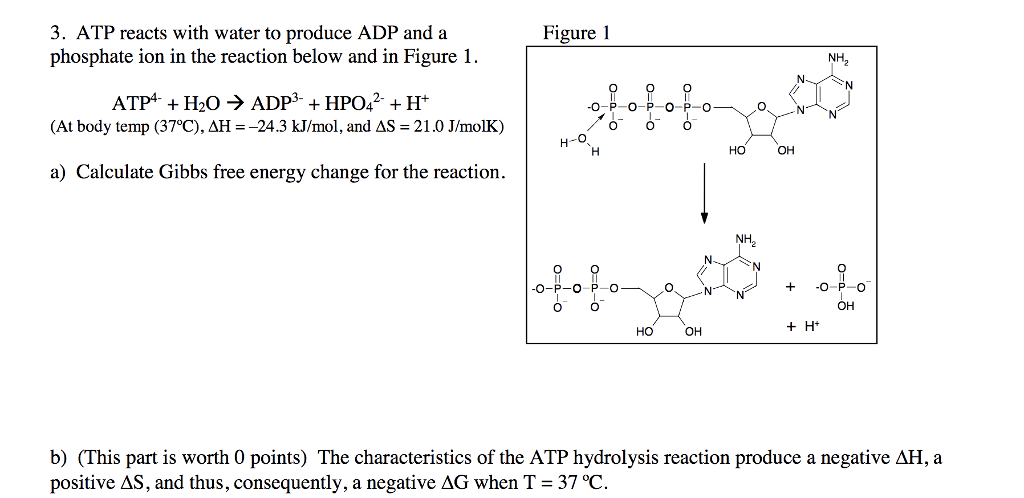
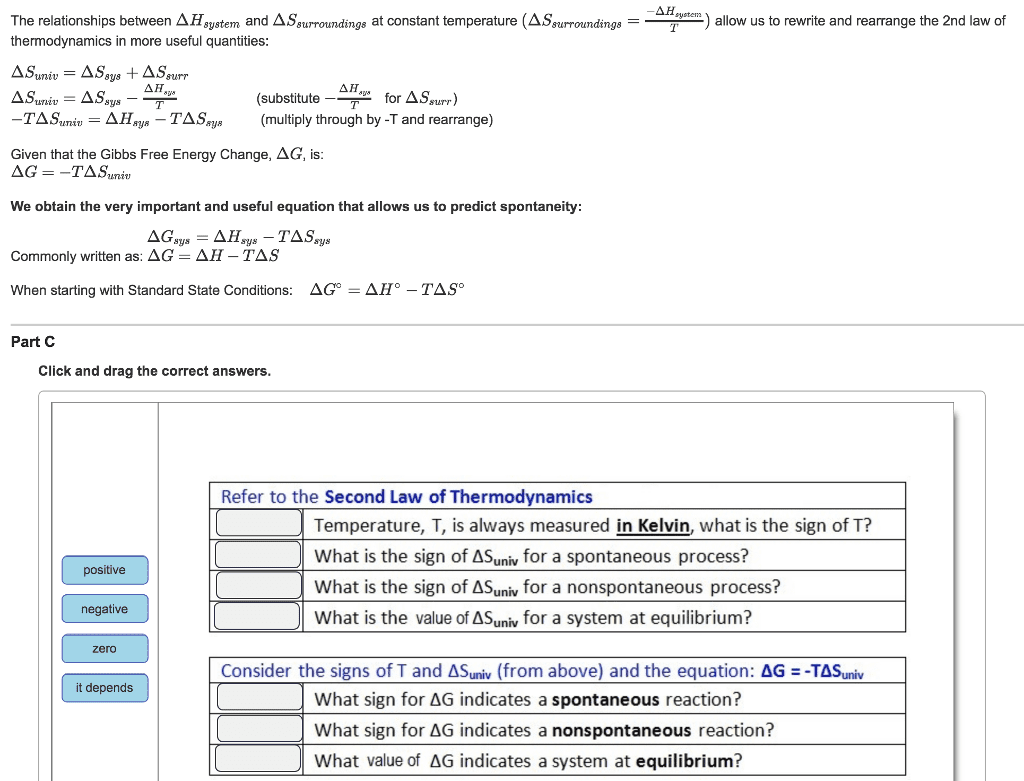
CHEM 223 Lecture 1: DOC.PDF
36 views4 pages
30 Sep 2016
School
Department
Course
Professor
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
1)
How are the equilibrium constant and the standard cell potential of an oxidation-reduction reaction related?
| a) They are not related. The equilibrium constant derives from enthalpy while reduction potential only deals with charge. |
| b) Both are a function of the standard change in Gibbs free energy. |
| c) Both are related to the amount of work that can be done by a reaction on its surroundings. |
| d) Both are a function of the number of electrons transferred in the reaction. 2) Calculate the standard cell potential (Eâ) for the reaction X(s)+Y+(aq)âX+(aq)+Y(s) if K = 2.30Ã10â4. Express your answer to three significant figures and include the appropriate units. 3) Consider the reaction Mg(s)+Fe2+(aq)âMg2+(aq)+Fe(s) at 77 âC , where [Fe2+]= 3.80 M and [Mg2+]= 0.310 M . A) What is the value for the temperature, T, in kelvins? Express your answer to three significant figures and include the appropriate units. B) What is the value for n? Express your answer as an integer and include the appropriate units (i.e. enter mol for moles) |