CHEM 212 Lecture Notes - Bohr Model, Atom, Actinide
Document Summary
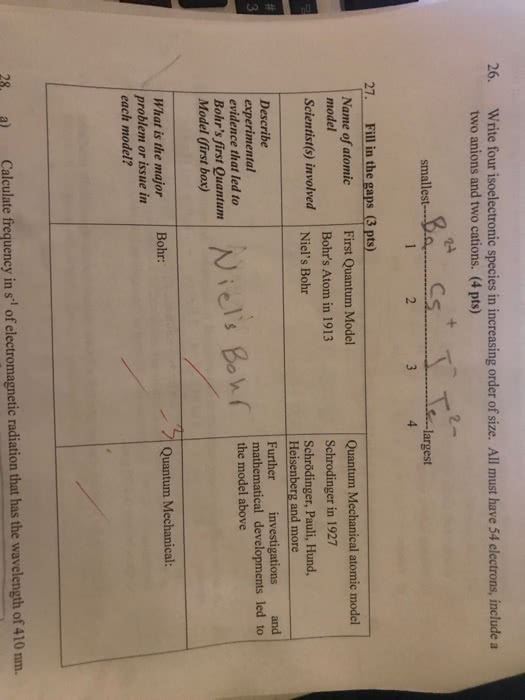
The theories of atomic and molecular structure depend on quantum mechanics to de- scribe atoms and molecules in mathematical terms. Although the details of quantum mechanics require considerable mathematical sophistication, it is possible to under- stand the principles involved with only a moderate amount of mathematics. This chap- ter presents the fundamentals needed to explain atomic and molecular structures in qualitative or semiquantitative terms. 2-1 although the greek philosophers democritus (460-370 bc) and epicurus (341-270. Hlstorlcal bc) presented views of nature that included atoms, many hundreds of years passed. Development of before experimental studies could establish the quantitative relationships needed for a. In 1808, john dalton published a new system of chemical. ~ h i l o s o ~ h ~ , " in which he proposed that the ultimate particles of all homogeneous bodies are perfectly alike in weight, figure, etc.