please be very specific
please be very specific
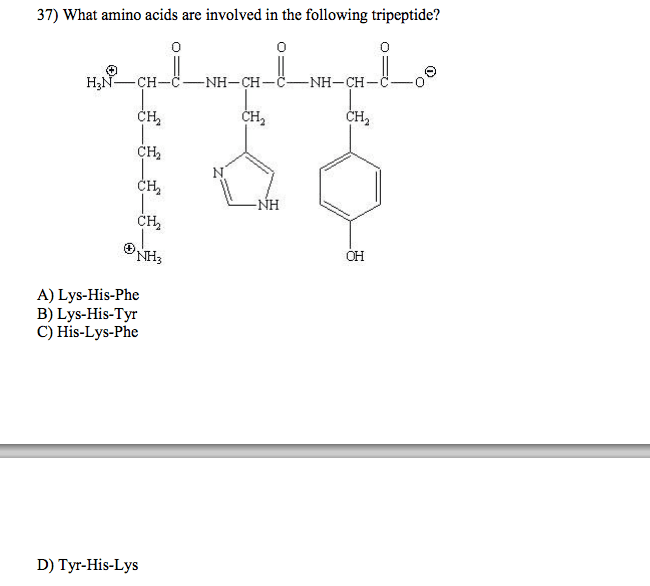
Draw the complete structure of the tetra-peptide ser-gln-glu-ala as you would expect it to exist in solution at pH 4.2 and calculate the net charge on the peptide at pH 4.2. Show all atoms including all hydrogen atoms and all bonds. You may use the abbreviations -CH2 and -CH3, but no others. Hint: alanine is at the C or carboxy terminus of the peptide. Indicate the charge of the most predominant form of each charged group. Structure at pH 4.2: one or more of the weak acid groups in the peptide will become deprotonated. What will be the net charge on this peptide at pH 11? Amide bonds are formed between amine and carboxylic acid functional (chemical) groups. Hydrolysis of amide bonds will give the free amine and carboxyl group. For the tetra-peptide you drew in 3a, how many amide (peptide) bonds in the peptide backbone can be hydrolyzed? Do any of the amino acid side-chains in this tetrapeptide have amide bonds? Circle one: YES NO If yes, Indicate which amino acid(s). If the tetra-peptide (above) is part of a larger water soluble protein, would you predict it to be found on the surface of the protein (interacting with the polar aqueous environment) or buried in the middle of the protein (interacting with hydrophobic amino acid side-chains)? Briefly explain.
Show transcribed image text Draw the complete structure of the tetra-peptide ser-gln-glu-ala as you would expect it to exist in solution at pH 4.2 and calculate the net charge on the peptide at pH 4.2. Show all atoms including all hydrogen atoms and all bonds. You may use the abbreviations -CH2 and -CH3, but no others. Hint: alanine is at the C or carboxy terminus of the peptide. Indicate the charge of the most predominant form of each charged group. Structure at pH 4.2: one or more of the weak acid groups in the peptide will become deprotonated. What will be the net charge on this peptide at pH 11? Amide bonds are formed between amine and carboxylic acid functional (chemical) groups. Hydrolysis of amide bonds will give the free amine and carboxyl group. For the tetra-peptide you drew in 3a, how many amide (peptide) bonds in the peptide backbone can be hydrolyzed? Do any of the amino acid side-chains in this tetrapeptide have amide bonds? Circle one: YES NO If yes, Indicate which amino acid(s). If the tetra-peptide (above) is part of a larger water soluble protein, would you predict it to be found on the surface of the protein (interacting with the polar aqueous environment) or buried in the middle of the protein (interacting with hydrophobic amino acid side-chains)? Briefly explain.