BIOCHEM 3H03 Lecture 26: BIOCHEM 3H03 LECTURE 26
Document Summary
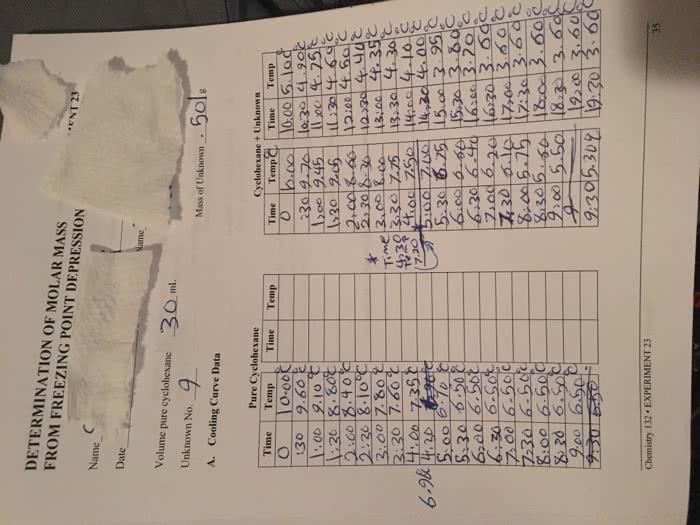
Analytical techniques: analytical test- osmolality o osmolality(plasmaandurine) o. Osmometry= technique for measuring the concentration of solutes that contribute to osmotic pressure of a solution o. Colligativeproperties=propertiesthatdependonnumberandnotthe nature of the dissolved particles ex. Osmotic pressure, elevation of boiling point, freezing point depression, vapor pressure lowering are all colligative properties: how to measure freezing point depression. Quickly cool the sample to below the freezing point (supercool, still in liquid form) Solution reaches equilibrium where freezing and thawing are equal accurately measure this temperature. Specific gravity uring refractometry, light will refract different with different densities* Couldthisbemethanolorethyleneglycolpoisoning?isthereanother osmotically active particle there that we are not calculating o. Calculatedosmolality,withequation=285mosmol o measured osmolality= 312 mosmol o there is a osmolar gap of 27, which suggests presence of abnormal osmole present o patient airlifted to hospital where [ethylene glycol] was found to be. Measurement of na and k flame emission spectroscopy: metalionswillemitlightofspecificwavelengthwhen given enough energy.