CHEM 1A03 Lecture Notes - Lecture 6: Lipophilicity, Partition Coefficient, Acid Dissociation Constant

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
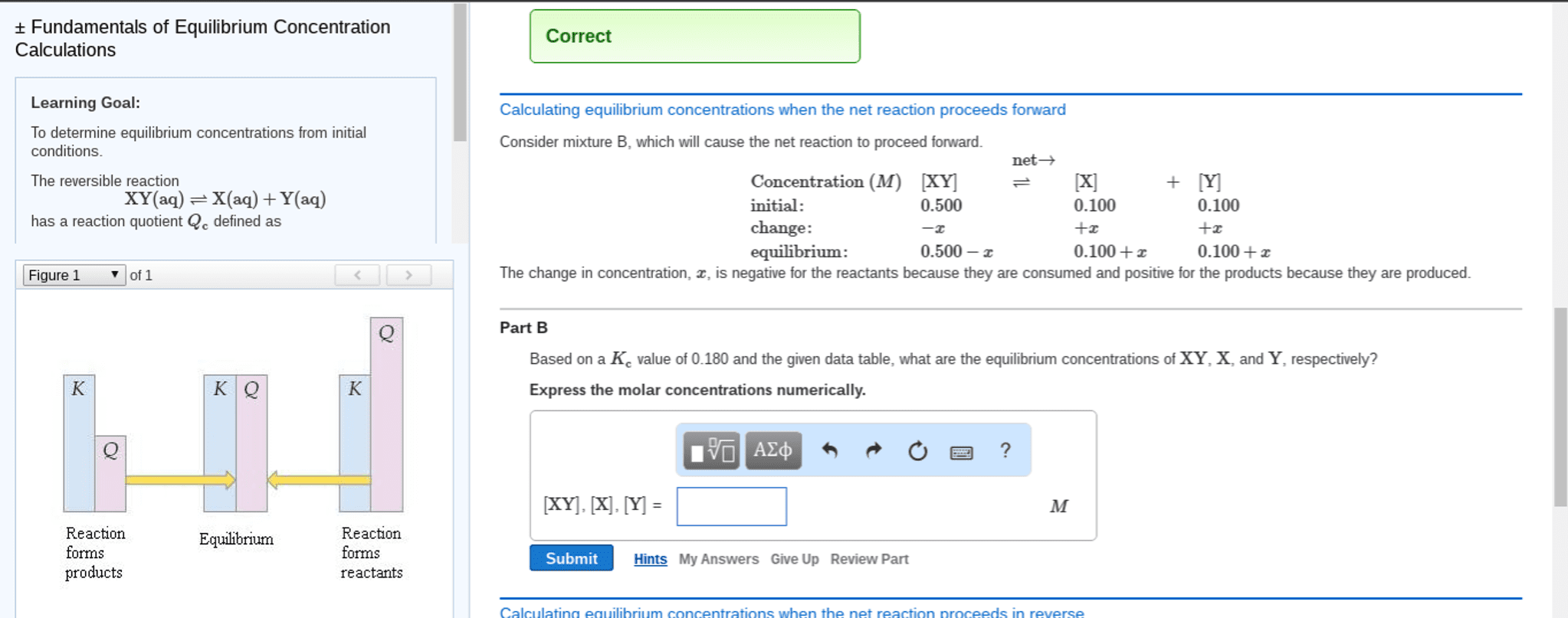
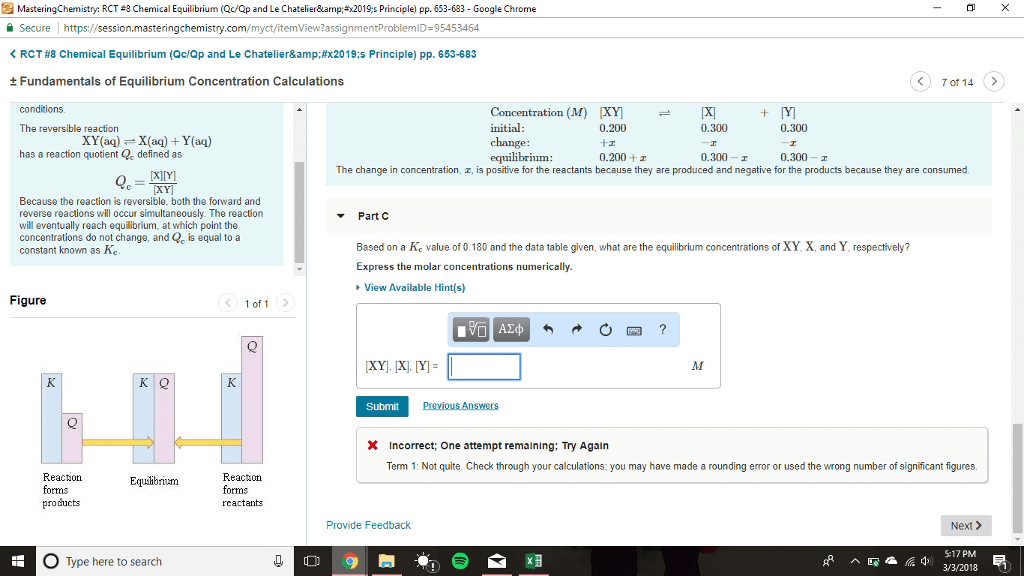
=(cid:4666)(cid:1829)(cid:3051)(cid:4667)(cid:4666)(cid:1830)(cid:3052)(cid:4667) (cid:4666)(cid:1827)(cid:3040)(cid:4667)(cid:4666)(cid:1828)(cid:3041)(cid:4667: q=reaction quotient no units, a=activity (concentration mols/l, a, b=reactants, c, d=products, m, n=coefficients of reactants, x, y=coefficients of products. The reaction quotient for the above reaction is q=[ca2+ They are both to the power of 1 since their coefficients are 1 but there is no need to write them. =(cid:4666)(cid:1829)(cid:3051)(cid:4667)(cid:4666)(cid:1830)(cid:3052)(cid:4667) (cid:4666)(cid:1827)(cid:3040)(cid:4667)(cid:4666)(cid:1828)(cid:3041)(cid:4667: k=reaction quotient at equilibrium no units, a=activity (concentration mols/l, a, b=reactants, c, d=products, m, n=coefficients of reactants, x, y=coefficients of products. *use partial pressures instead of concentrations for gases* If k is large, the numerator is higher meaning there are more products being made: the reaction is moving towards the right. If k is small, the denominator is higher meaning more reactants are being made: the reaction is moving towards the left, k is dependents on ph, temperature, and pressure. If a solute is added to this, it will be partitioned: partitioned: equilibrium distribution of a solute between two layers.