CHEM 1A03 Lecture Notes - Lecture 7: Equilibrium Constant, Dynamic Equilibrium, Rice Chart

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
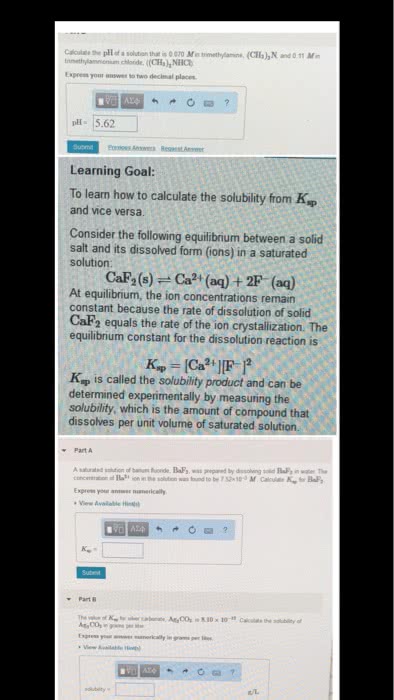
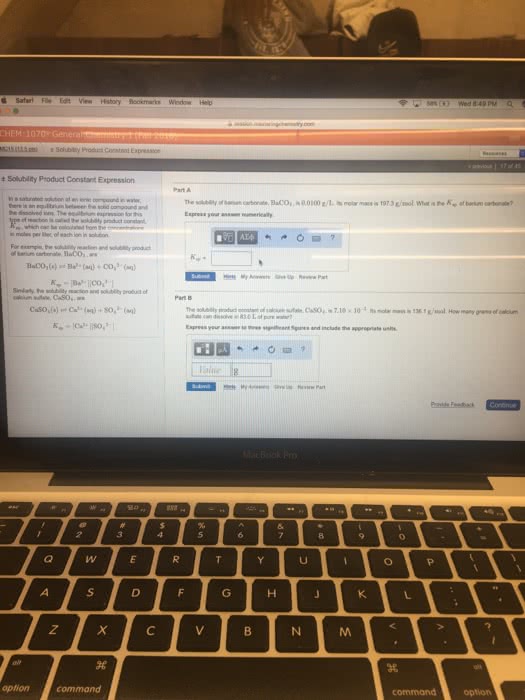
Solubility is the maximum amount of the substance, which will dissolve at a given temperature (at this point the solution is saturated) A saturate solution is a solution in which dynamic equilibrium exists between undissolved and the dissolved solute. A substance is considered soluble if it will form a solution of concentration. > 0. 1 moles per litre at room temperature otherwise it is insoluble. A low value of ksp means the concentrations of ions are low at equilibrium. When a solid electrolyte is in a state of equilibrium with a saturated solution the concentration of a solid is a constant. The equilibrium constant is called solubility product constant. This new constant, equal to the products of the concentrations of all the ions present is called solubility product. Example1: copper (1) bromide has a measured solubility of 2. 0 x 10-4 mol/l at. = 4. 0 x 10-8 (there is no unit) Example 2: calculate the ksp value for bismuth sulfide (bi2s3(s)).