CHEM 281 Lecture Notes - Lecture 4: Molecular Orbital Diagram, Substituent, Aromaticity
Document Summary
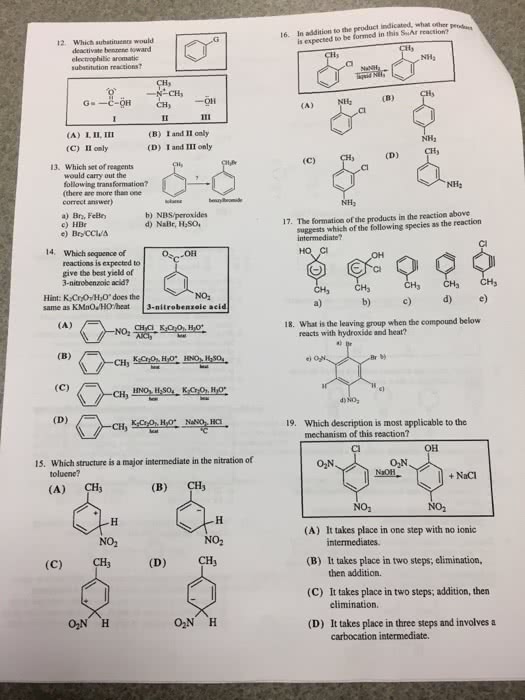
For only the aromatic molecules below, so how many -electrons are present in each aromatic system. Using frost-musulin diagrams, and only 4 aromatic molecules above, generate the relative frontier molecular orbital diagram for your choices. Nr2ohsroronhororrarxoronr2oorcnso3hcx3no2nr3substituentrate e ectregiochemistrystrongly activatingortho, paramoderately activatingortho, paraweakly activatingortho, paraweakly deactivatingortho, paramoderately deactivatingmetastrongly deactivatingmetashow the mechanism for each of the electrophilic aromatic substitutions below. Deduce the identify of the structure with the molecular formula c8h7no that exhibits the following ir, 1h nmr and 13c nmr spectra. Deduce the identify of the structure with the molecular formula c9h9no3 that exhibits the following ir, 1h nmr and 13c nmr spectra. Deduce the identify of the structure with the molecular formula c9h11br that exhibits the following ir, 1h nmr and 13c nmr spectra. Deduce the identify of the structure with the molecular formula c11h17no that exhibits the following ir, 1h nmr and 13c nmr spectra.