CHY 103 Lecture Notes - Lecture 2: Reagent, Chemical Equation
Document Summary
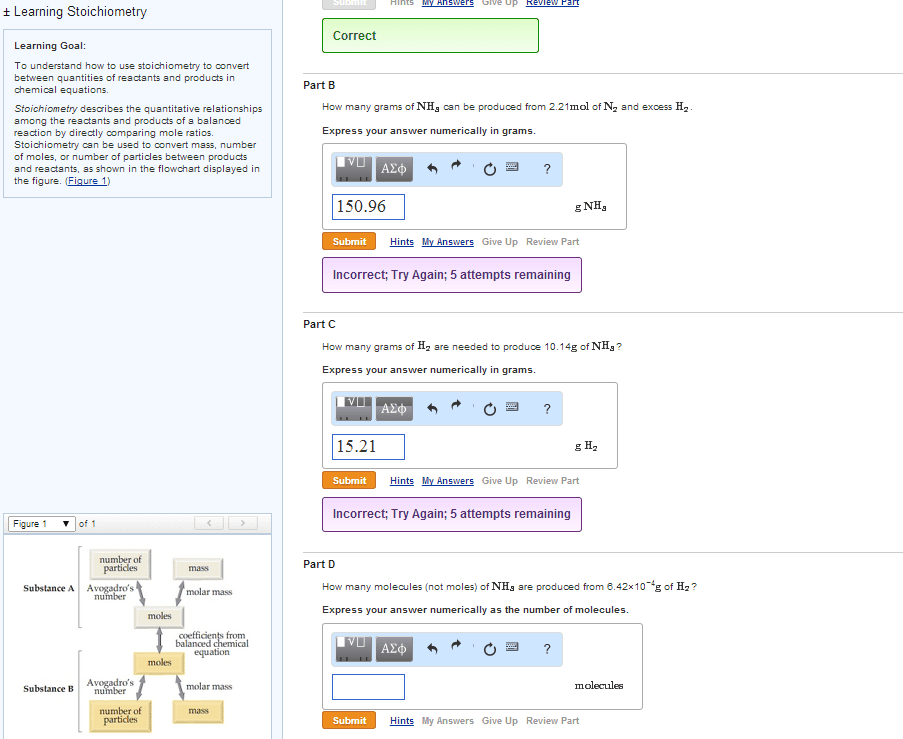
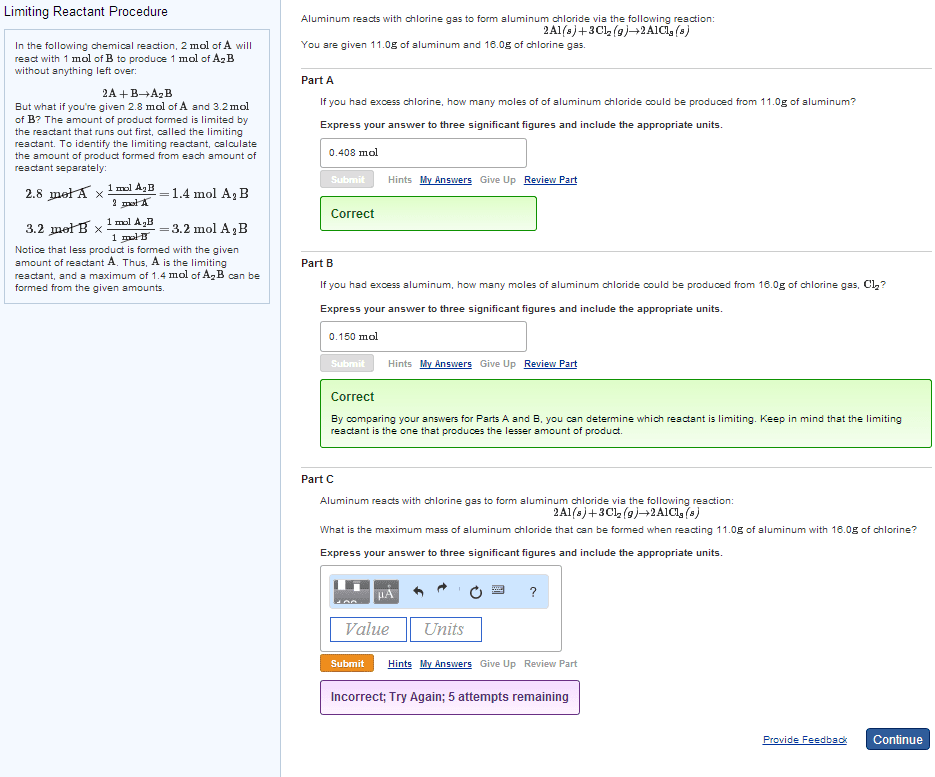
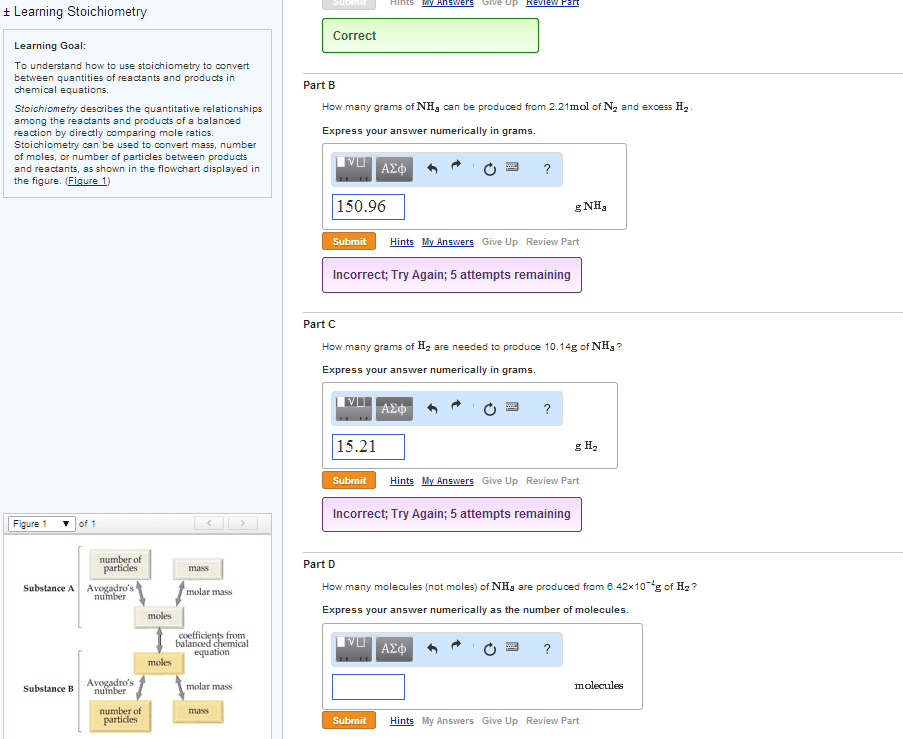
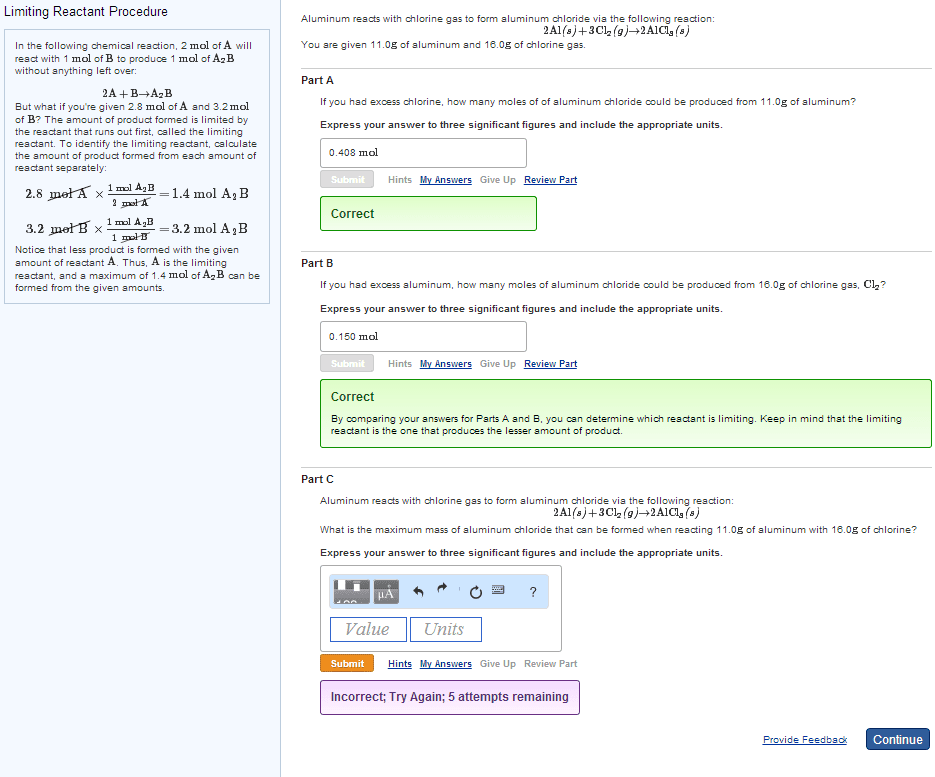
The coefficients of a chemical equation relate the amount of each substance in the equation to the other substances in the equation. They can also be used to predict how much product can be produced or how much reactant is needed. Procedure for stoichiometry problems: convert grams of substance a to moles of substance a, convert moles of substance a to moles of substance b using stoichiometric ratio and balanced equation, convert moles back into grams for desired product. 5ki + kio3 + 6hno3 6kno3 + 3i2 +3h2o. What mass of i2 is produced if 13. 10 g ki is reacted with excess kio3 and hno3 ? data: m(ki) = 166. 0 g mol-1 , m(i2) = 253. 8 g mol-1. No undue complications so far - we were told that we had excess of the other reagents. In general, unless we are told that the other reagents are in excess, we must never assume it.