CHEM 121 Lecture 13: 66Lecture 13.pdf
35 views23 pages
19 Jun 2013
School
Department
Course
Professor
Document Summary
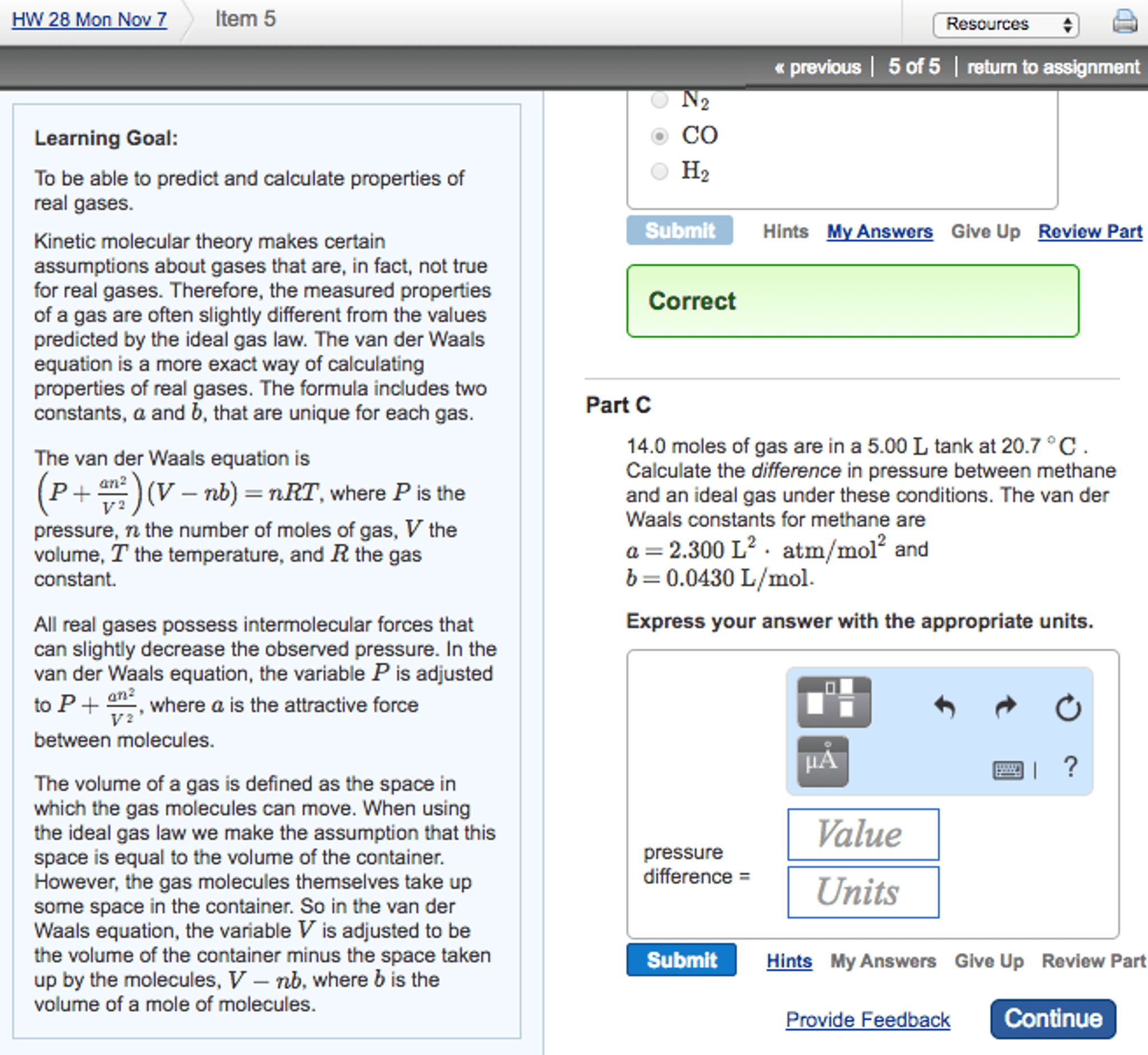
Energy and enthalpy of heating a gas (cid:96) at constant volume: qv = ncv t (cv = 3/2 r) E = cv t per mole or e = nc v t for n moles. For any ideal gas the change in internal energy is ncv t. Energy and enthalpy of heating a gas (cont"d) (cid:96) at constant pressure: qp = ncp t. E = qp + w = qp - p v qp = e + p v. E for an ideal gas depends only on t (not on p or v) T when an ideal gas is heated whether at. E = nc v since pv = nrt then: h = e + (nrt) H = e + (pv) and e = ncv t then: h = ncv t + nr t since cp = cv+ r. H = nc p t for constant volume or constant pressure.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232