CHEM 122 Lecture Notes - Lecture 9: Ethylene, Joule, Thermodynamics
Document Summary
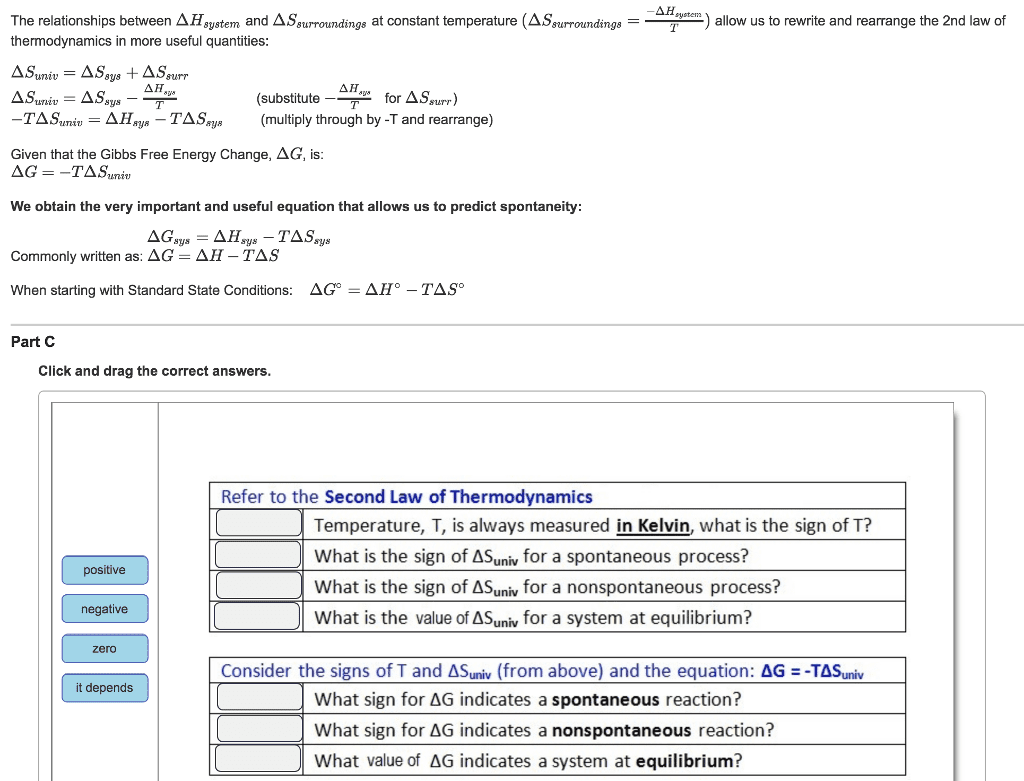
Chem 122: general chemistry ii - lecture 9-1: effect of temperature and spontaneity (10. 5, 10. 6, and 10. 8) Consider the change of state of water from liquid solid: H2o(l) h2o(s) exothermic process (heat is released to form solid) We would expect the entropy change for this system to be negative since we are going from a liquid solid (i. e. based on positional probability) Yet, we know this process is spontaneous at temperature below 0 c we must consider the entropy change of the system ( ssys ) and the entropy change of the surroundings ( ssurr. ) and the relative effect the temperature has on the importance of these two terms. Suniverse = ssys + ssurr. If the suniverse is greater than zero then the process is spontaneous in the direction written. If the suniverse is less than zero then the process is not spontaneous in the direction written or is spontaneous in the opposite direction.