CHEM 1010H Lecture Notes - Lecture 4: Reaction Rate, Dd National, Rate Equation
Document Summary
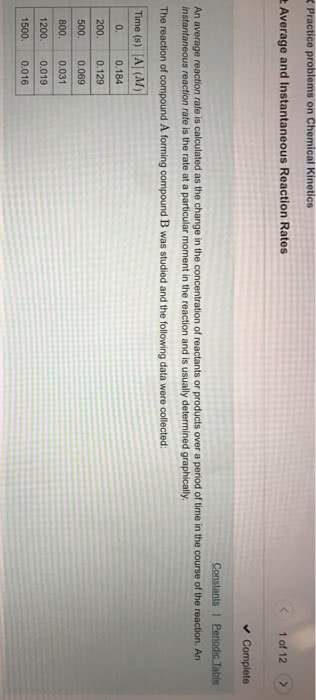
Rate: a measure of how something changes with time the rate of decrease of [reactants] per unit time. The rate of increase of [products] per unit time. The laws that govern chemical reaction rates. The theories that we use to explain the laws. This is what we quantify with a reaction rate. Measuring chemical rates: h2 reacts with i2 to give hi, a strong acid: = [(cid:3009)(cid:2870)] measure the decrease in the [reactant] over time. = (cid:883)[(cid:1834)(cid:1835)] (cid:884) measure the increase in [product] over time. The reaction rate is the same in all cases ( all equal ) only difference is the species (h2, For the reaction aa + bb (cid:3450) cc + dd. To measure concentration changes, use a property of one of the. Species in ou reaction that changes proportionally to its concentration and is easily measured. You can comply that change to the number of moles generated.