CHEM101 Lecture Notes - Lecture 11: Formal Charge, Logarithmic Scale, Chemical Formula

77
CHEM101 Full Course Notes
Verified Note
77 documents
Document Summary
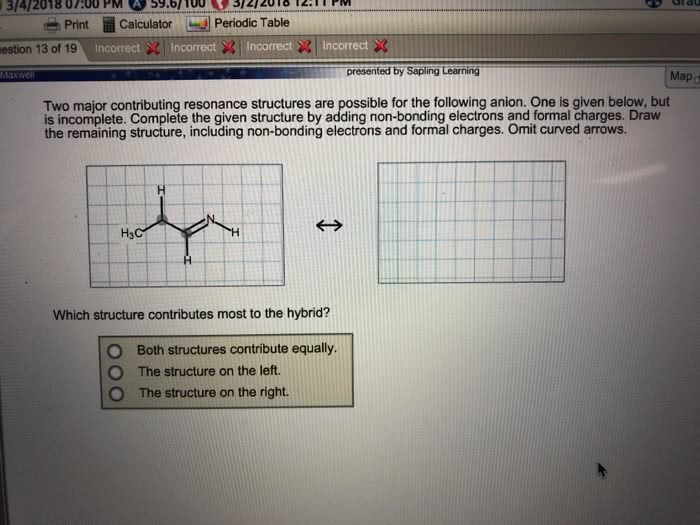
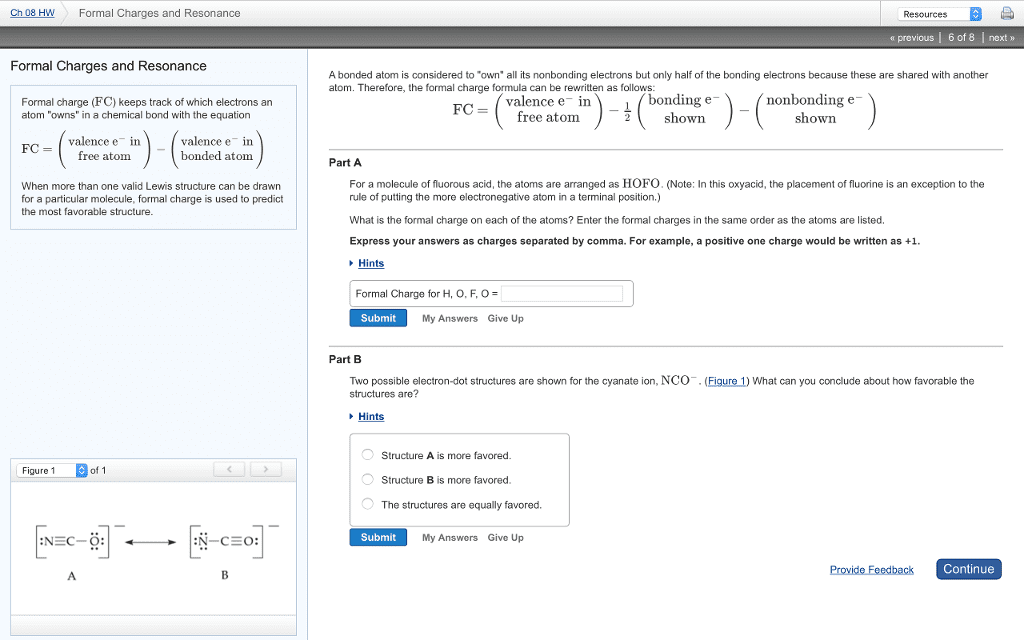
Resonance, resonance structure occurs when you can draw a lewis structure of a molecule in multiple ways. In this case, bond order is equal to the # of bonding electrons / # of bonds: to figure out which resonance structure is correct you need to look at the following criteria. Reasoning is because positive charges are formed on the most electronegative atoms which is undesirable. This usually results when using fluorine and chlorine: sometimes molecules will have odd numbered electrons, meaning it does not complete the octet as well. Acid is a compound containing h in the formula and yields h3o+ Base is a compound containing oh in the formula and yields oh- Neutralization reaction yields water and an ionic salt. However, this has limits such as the equations must be in aqueous solution and some bases do not contain oh in the chemical formula but still yield oh: the bronsted-lowry definition.