CHEM101 Lecture Notes - Lecture 32: Kinetic Theory Of Gases, Ideal Gas Law, Molar Mass
CHEM101 verified notes
32/41View all
Document Summary
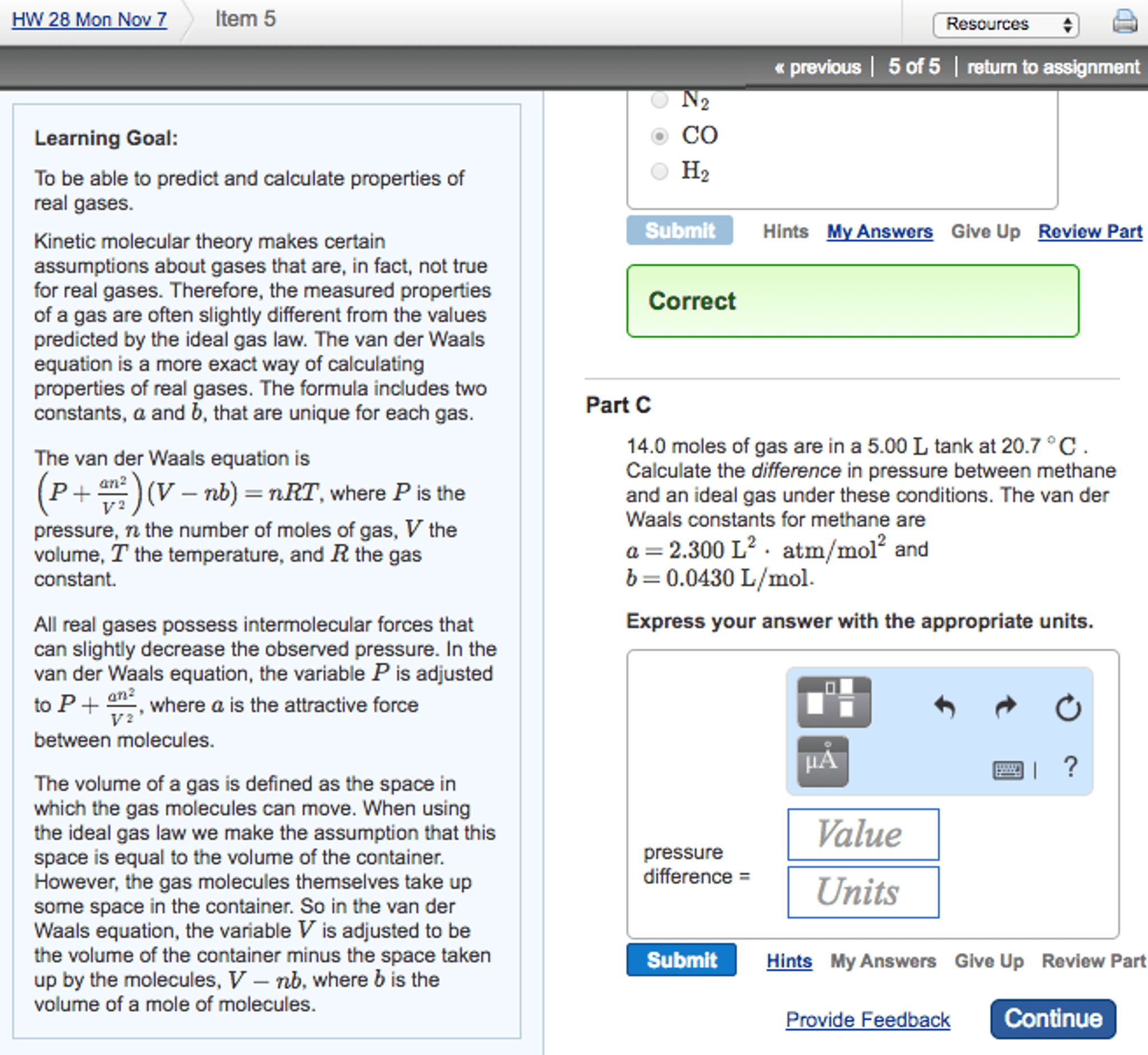
March 20: gases and kinetic molecular theory, measuring quantities of gases, ideal gas. There are five unique behaviours of gases that are different to those of solids and liquids: volume changes a lot depending on temperature. The volume is the space that the molecules occupy in their gas phase. Whereas solids or liquids are measured in grams/ml: gases form solutions at any proportion, meaning they will mix together in any ratio. This is because the gas molecules don"t interact with one another. Stoichiometry is when the number of moles of gas is related to the mass of gas. Mass = molar mass x mols = 28 g/mol x 15 mol = 420g volume = approx. When large masses are used there are also large volumes, and that volume has to be kept in a large container, which has its own mass.