CHEM101 Lecture Notes - Lecture 26: Mean Free Path, Ionic Radius, Intermolecular Force

77
CHEM101 Full Course Notes
Verified Note
77 documents
Document Summary
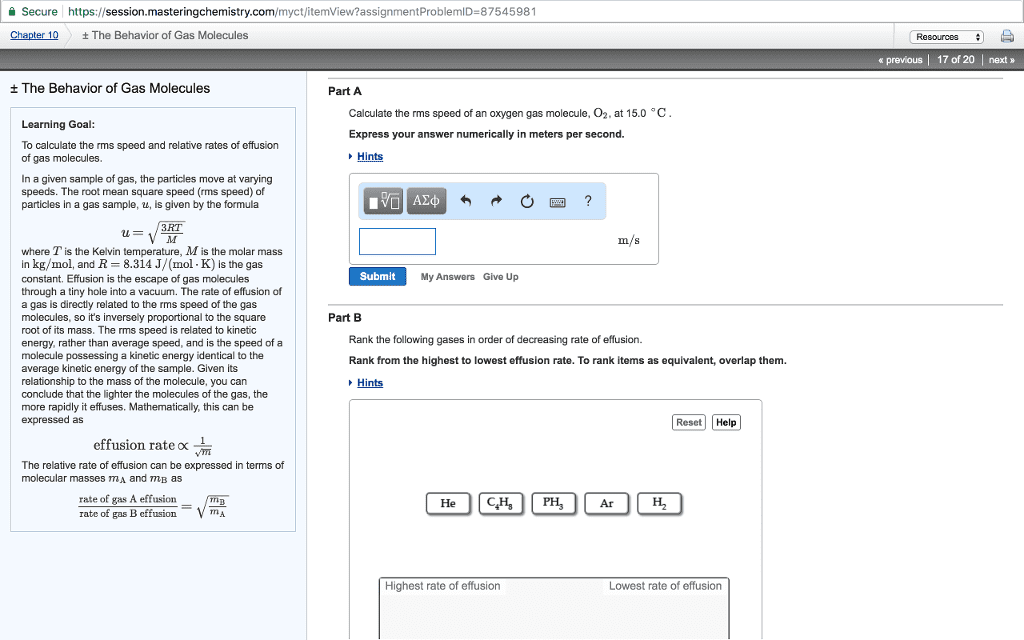
Efusion and difusion: process of a gas molecule escaping through a small hole, graham"s law of efusion. How many gas molecules / second pass through the hole/barrier. At the same temperature we know that lighter gas molecules move faster than heavy gas molecules. Rate of efusion of gas a / rate of efusion of gas b = sqrt [mgasb/mgasa] At the same temperature in the same volume at the same pressure etc compare the rate of efusion for helium and o2 sqrt [32/2] = sqrt [16] = 4 helium will efuse 4x faster than o2: difusion. Net movement of gas molecules (or other molecules) from an area of high concentraion (high pressure) to an area of low concentraion (low pressure) Rate of difusion = rodgasa/rodgasb = sqrt [mb / ma] Mean free path: average distance molecules travel before a collision. It is very small and moves very quickly. Collision frequency = speed / mean free path.