CHEM103 Lecture Notes - Lecture 1: Mass Number, Carbon-12, Atomic Number
51 views1 pages
3 May 2016
School
Department
Course
Professor

128
CHEM103 Full Course Notes
Verified Note
128 documents
Document Summary
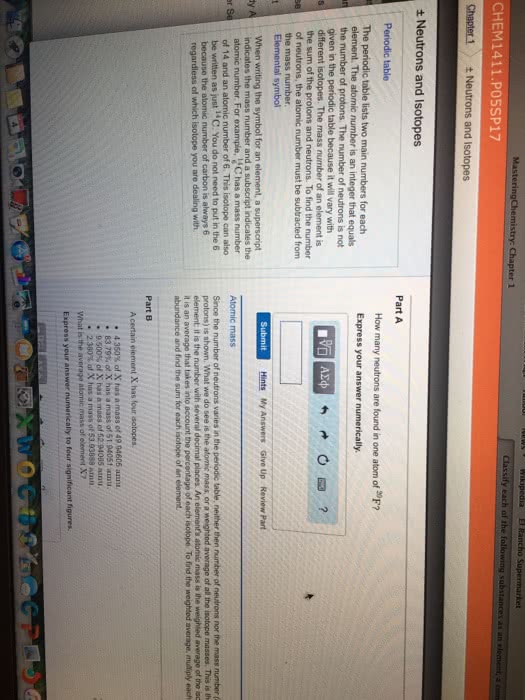
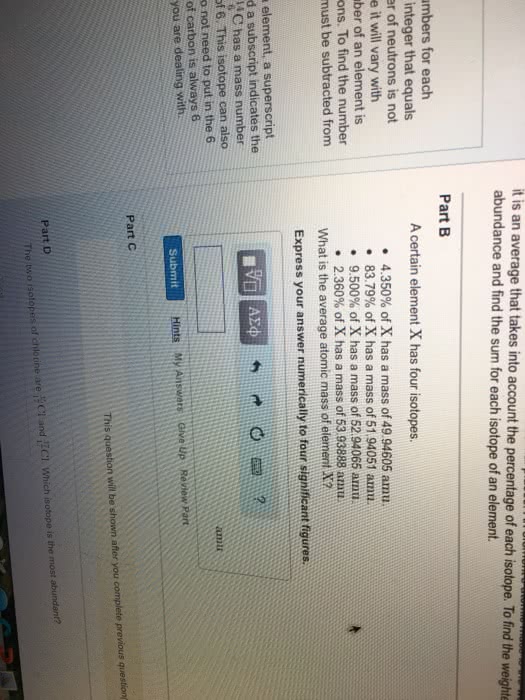
Mass number total number of protons and neutrons. Isotopes are the same element with different numbers of neutrons. Atomic symbol derived from element names in english, latin, and greek. Number of neutrons can be determined from subtracting the atomic number from the mass number of the element. One periodic table element square is in the following format: Isotopes of elements have different numbers of neutrons and therefore have different masses. Artificial isotopes are isotopes which do not occur naturally and are seen in nuclear decay or prepared in particle accelerators: carbon 9, 10, 11, 15, and 16 are such artificial isotopes. There are three natural isotopes of silicon: silicon 28 14 neutrons, silicon 29 15 neutrons, silicon 30 16 neutrons.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232