CHEM261 Lecture Notes - Infrared Spectroscopy, Wavenumber, Transmittance

1
CHEM261 Full Course Notes
Verified Note
1 document
Document Summary
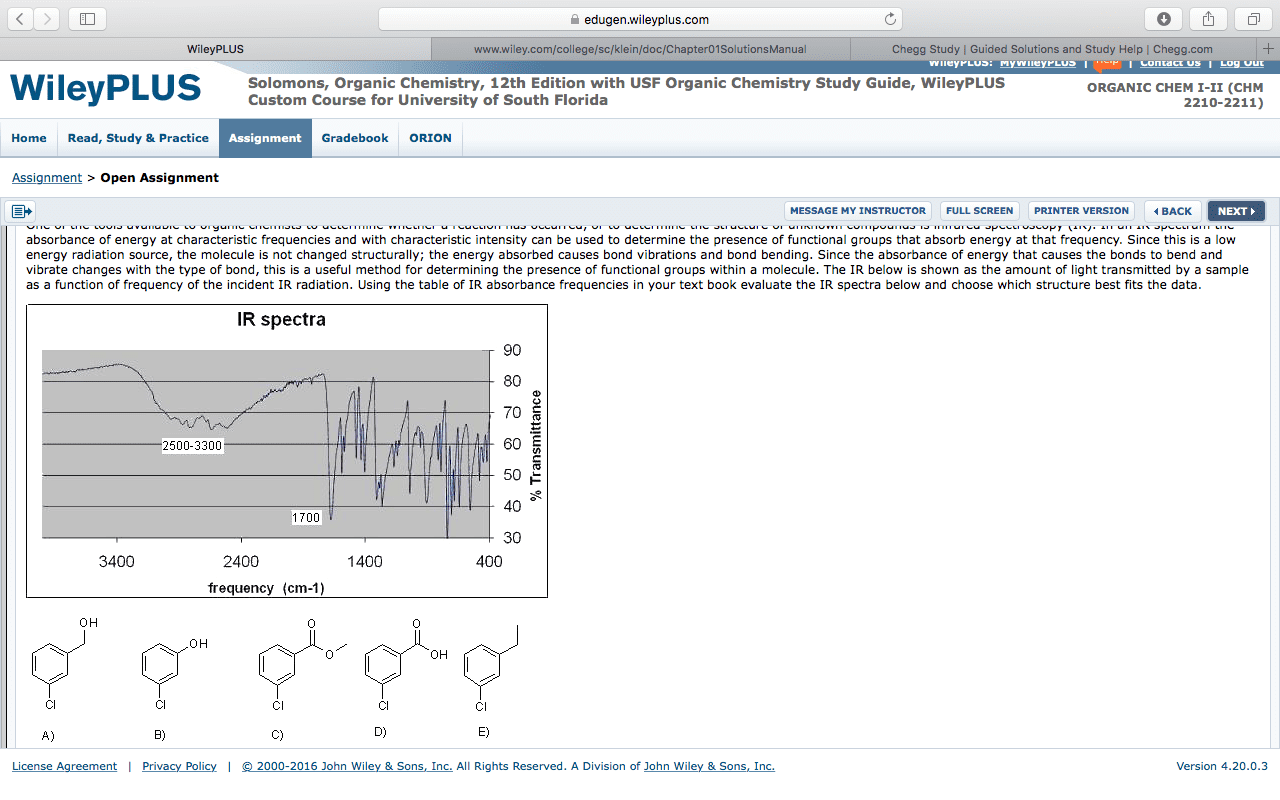
Infrared spectroscopy involves studying the interaction of infrared radiation on atoms or molecules. When a sample of an organic compound is subjected to infrared radiation the compounds absorbs some of this energy. As energy at certain wavelengths is absorbed a detector can be used to record the decrease in intensity of the radiation as it passes through the sample. An absorption spectrum is produced showing the percentage transmittance versus the wavenumber. A 95% transmittance means that most radiation at a particular frequency passed straight through the sample. A 5% transmittance means that nearly all the radiation at a particular frequency was absorbed by the sample. The amount of absorption or transmittance of infrared radiation provides information about the bonds in the sample. If a particular frequency is being absorbed as it passes through a sample then it must mean that its energy is being transferred to the compound.