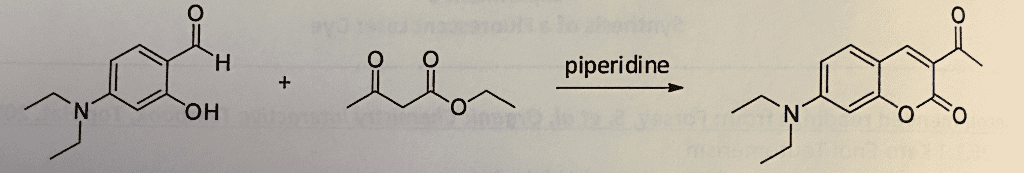
CHEM263 Lecture Notes - Lecture 9: Carbonyl Group, Nucleophilic Conjugate Addition, Enol
Document Summary
Unit 7: selectivity and reactivity in enolate reactions. , -unsaturated carbonyls are ambident electrophiles that can undergo either direct 1,2-addition or conjugate 1,4-addition. An ambident electrophile is a functional group with two electrophilic sites. Unit 7_selectivity and reactivity in enolate react page 1. The two electrophilic carbons in , -unsaturated carbonyls have different reactivity. Nucleophiles will preferentially react with one site than the other based on charge density. Unit 7_selectivity and reactivity in enolate react page 2. Kinetics are governed by the activation energy and dominate when reactions are irreversible. Thermodynamics are governed by the free energy change between reactants and products and dominate when reactions are reversible. A reaction that is under kinetic control uses reaction conditions that favor the product that is most quickly formed. When a reaction is reversible, the products formed depend on their relative stability. This type of reaction is under thermodynamic control. Unit 7_selectivity and reactivity in enolate react page 3.