RELIG220 Lecture : The Sunna and Hadith
Get access


Related Documents
Related Questions
Adam is a part-time employee who earned $495.00 during the mostrecent pay period. He is married with two withholding allowances.Prior to this pay period, his year-to-date pay is $6,492.39. Howmuch should be withheld from Adam's gross pay for Social Securitytax for the most recent period, assuming no pre-tax deductions?
$40.02
$30.69
$37.92
$28.46
Julian is a part-time, nonexempt employee in Texas who earns$21.50 per hour. During the last biweekly pay period he worked 45hours. He is single with one withholding allowance (use thewage-bracket table). What is his net pay? (Donot roundinterim calculations,only round finalanswer to twodecimal points.)
Wage Bracket Method Tables for Income TaxWithholding | ||||||||||||
And the wages are | And the number of withholding allowances claimedis- | |||||||||||
Atleast | But less | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
The amount of income tax to be withheld is- | ||||||||||||
$800 | $820 | $90 | $67 | $44 | $26 | $11 | $0 | $0 | $0 | $0 | $0 | $0 |
820 | 840 | 93 | 70 | 47 | 28 | 13 | 0 | 0 | 0 | 0 | 0 | 0 |
840 | 860 | 96 | 73 | 50 | 30 | 15 | 0 | 0 | 0 | 0 | 0 | 0 |
860 | 880 | 99 | 76 | 53 | 32 | 17 | 1 | 0 | 0 | 0 | 0 | 0 |
880 | 900 | 102 | 79 | 56 | 34 | 19 | 3 | 0 | 0 | 0 | 0 | 0 |
900 | 920 | 105 | 82 | 59 | 36 | 21 | 5 | 0 | 0 | 0 | 0 | 0 |
920 | 940 | 108 | 85 | 62 | 39 | 23 | 7 | 0 | 0 | 0 | 0 | 0 |
940 | 960 | 111 | 88 | 65 | 42 | 25 | 9 | 0 | 0 | 0 | 0 | 0 |
960 | 980 | 114 | 91 | 68 | 45 | 27 | 11 | 0 | 0 | 0 | 0 | 0 |
980 | 1,000 | 117 | 94 | 71 | 48 | 29 | 13 | 0 | 0 | 0 | 0 | 0 |
1,000 | 1,020 | 120 | 97 | 74 | 51 | 31 | 15 | 0 | 0 | 0 | 0 | 0 |
1,020 | 1,040 | 123 | 100 | 77 | 54 | 33 | 17 | 2 | 0 | 0 | 0 | 0 |
1,040 | 1,060 | 126 | 103 | 80 | 57 | 35 | 19 | 4 | 0 | 0 | 0 | 0 |
1,060 | 1,080 | 129 | 106 | 83 | 60 | 37 | 21 | 6 | 0 | 0 | 0 | 0 |
1,080 | 1,100 | 132 | 109 | 86 | 63 | 40 | 23 | 8 | 0 | 0 | 0 | 0 |
1,100 | 1,120 | 135 | 112 | 89 | 66 | 43 | 25 | 10 | 0 | 0 | 0 | 0 |
1,120 | 1,140 | 138 | 115 | 92 | 69 | 46 | 27 | 12 | 0 | 0 | 0 | 0 |
1,140 | 1,160 | 141 | 118 | 95 | 72 | 49 | 29 | 14 | 0 | 0 | 0 | 0 |
1,160 | 1,180 | 144 | 121 | 98 | 75 | 52 | 31 | 16 | 0 | 0 | 0 | 0 |
1,180 | 1,200 | 147 | 124 | 101 | 78 | 55 | 33 | 18 | 2 | 0 | 0 | 0 |
1,200 | 1,220 | 150 | 127 | 104 | 81 | 58 | 35 | 20 | 4 | 0 | 0 | 0 |
1,220 | 1,240 | 153 | 130 | 107 | 84 | 61 | 38 | 22 | 6 | 0 | 0 | 0 |
1,240 | 1,260 | 156 | 133 | 110 | 87 | 64 | 41 | 24 | 8 | 0 | 0 | 0 |
1,260 | 1,280 | 159 | 136 | 113 | 90 | 67 | 44 | 26 | 10 | 0 | 0 | 0 |
1,280 | 1,300 | 162 | 139 | 116 | 93 | 70 | 47 | 28 | 12 | 0 | 0 | 0 |
1,300 | 1,320 | 165 | 142 | 119 | 96 | 73 | 50 | 30 | 14 | 0 | 0 | 0 |
1,320 | 1,340 | 168 | 145 | 122 | 99 | 76 | 53 | 32 | 16 | 1 | 0 | 0 |
1,340 | 1,360 | 171 | 148 | 125 | 102 | 79 | 56 | 34 | 18 | 3 | 0 | 0 |
1,360 | 1,380 | 174 | 151 | 128 | 105 | 82 | 59 | 36 | 20 | 5 | 0 | 0 |
1,380 | 1,400 | 177 | 154 | 131 | 108 | 85 | 62 | 39 | 22 | 7 | 0 | 0 |
1,400 | 1,420 | 180 | 157 | 134 | 111 | 88 | 65 | 42 | 24 | 9 | 0 | 0 |
1,420 | 1,440 | 183 | 160 | 137 | 114 | 91 | 68 | 45 | 26 | 11 | 0 | 0 |
1,440 | 1,460 | 186 | 163 | 140 | 117 | 94 | 71 | 48 | 28 | 13 | 0 | 0 |
1,460 | 1,480 | 189 | 166 | 143 | 120 | 97 | 74 | 51 | 30 | 15 | 0 | 0 |
1,480 | 1,500 | 192 | 169 | 146 | 123 | 100 | 77 | 54 | 32 | 17 | 2 | 0 |
1,500 | 1,520 | 195 | 172 | 149 | 126 | 103 | 80 | 57 | 34 | 19 | 4 | 0 |
1,520 | 1,540 | 199 | 175 | 152 | 129 | 106 | 83 | 60 | 37 | 21 | 6 | 0 |
1,540 | 1,560 | 204 | 178 | 155 | 132 | 109 | 86 | 63 | 40 | 23 | 8 | 0 |
1,560 | 1,580 | 209 | 181 | 158 | 135 | 112 | 89 | 66 | 43 | 25 | 10 | 0 |
1,580 | 1,600 | 214 | 184 | 161 | 138 | 115 | 92 | 69 | 46 | 27 | 12 | 0 |
1,600 | 1,620 | 219 | 187 | 164 | 141 | 118 | 95 | 72 | 49 | 29 | 14 | 0 |
1,620 | 1,640 | 224 | 190 | 167 | 144 | 121 | 98 | 75 | 52 | 31 | 16 | 0 |
1,640 | 1,660 | 229 | 193 | 170 | 147 | 124 | 101 | 78 | 55 | 33 | 18 | 2 |
1,660 | 1,680 | 234 | 196 | 173 | 150 | 127 | 104 | 81 | 58 | 35 | 20 | 4 |
1,680 | 1,700 | 239 | 200 | 176 | 153 | 130 | 107 | 84 | 61 | 38 | 22 | 6 |
1,700 | 1,720 | 244 | 205 | 179 | 156 | 133 | 110 | 87 | 64 | 41 | 24 | 8 |
1,720 | 1,740 | 249 | 210 | 182 | 159 | 136 | 113 | 90 | 67 | 44 | 26 | 10 |
1,740 | 1,760 | 254 | 215 | 185 | 162 | 139 | 116 | 93 | 70 | 47 | 28 | 12 |
1,760 | 1,780 | 259 | 220 | 188 | 165 | 142 | 119 | 96 | 73 | 50 | 30 | 14 |
1,780 | 1,800 | 264 | 225 | 191 | 168 | 145 | 122 | 99 | 76 | 53 | 32 | 16 |
1,800 | 1,820 | 269 | 230 | 194 | 171 | 148 | 125 | 102 | 79 | 56 | 34 | 18 |
1,820 | 1,840 | 274 | 235 | 197 | 174 | 151 | 128 | 105 | 82 | 59 | 36 | 20 |
1,840 | 1,860 | 279 | 240 | 202 | 177 | 154 | 131 | 108 | 85 | 62 | 39 | 22 |
1,860 | 1,880 | 284 | 245 | 207 | 180 | 157 | 134 | 111 | 88 | 65 | 42 | 24 |
1,880 | 1,900 | 289 | 250 | 212 | 183 | 160 | 137 | 114 | 91 | 68 | 45 | 26 |
1,900 | 1,920 | 294 | 255 | 217 | 186 | 163 | 140 | 117 | 94 | 71 | 48 | 28 |
1,920 | 1,940 | 299 | 260 | 222 | 189 | 166 | 143 | 120 | 97 | 74 | 51 | 30 |
1,940 | 1,960 | 304 | 265 | 227 | 192 | 169 | 146 | 123 | 100 | 77 | 54 | 32 |
1,960 | 1,980 | 309 | 270 | 232 | 195 | 172 | 149 | 126 | 103 | 80 | 57 | 34 |
1,980 | 2,000 | 314 | 275 | 237 | 198 | 175 | 152 | 129 | 106 | 83 | 60 | 37 |
2,000 | 2,020 | 319 | 280 | 242 | 203 | 178 | 155 | 132 | 109 | 86 | 63 | 40 |
2,020 | 2,040 | 324 | 285 | 247 | 208 | 181 | 158 | 135 | 112 | 89 | 66 | 43 |
2,040 | 2,060 | 329 | 290 | 252 | 213 | 184 | 161 | 138 | 115 | 92 | 69 | 46 |
2,060 | 2,080 | 334 | 295 | 257 | 218 | 187 | 164 | 141 | 118 | 95 | 72 | 49 |
2,080 | 2,100 | 339 | 300 | 262 | 223 | 190 | 167 | 144 | 121 | 98 | 75 | 52 |
$2,100 and over | Use Table 2(a) for a SINGLE person on page 45. Also see theinstructions on page 43. | |||||||||||
Page 52 | Publication 15 (2015) | |||||||||||
$818.40
$797.18
$825.99
$802.49
I need these ratios:
Return on Assets
|
Give It Some Thought 6.16
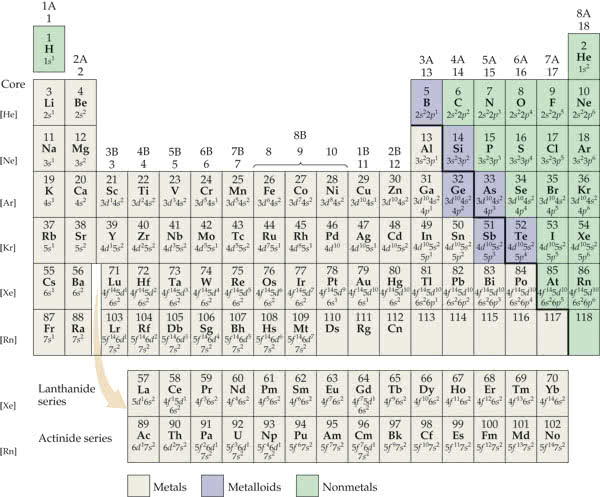
Valence electron configurations of the elements.
Part A
The elements Ni, Pd, and Pt are all in the same group. By examining the electron configurations for these elements in the following figure, what can you conclude about the relative energies of the nd and (n+1)s orbitals for this group?
The elements Ni, Pd, and Pt are all in the same group. By examining the electron configurations for these elements in the following figure, what can you conclude about the relative energies of the and orbitals for this group?
| nd > (n+1)s |
| nd < (n+1)s |
| nd = (n+1)s |
| It is impossible to conclude. |
