CHEM 3750 Lecture 1: Chapter1
155 views59 pages
24 Aug 2016
School
Department
Course
Professor
Document Summary
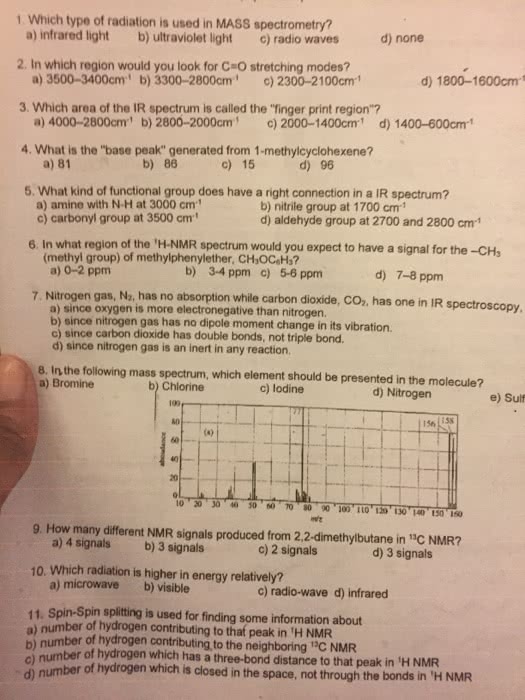
Atomic nuclei with odd mass numbers have angular momentum and behave as though they were spinning on an axis, that is, they behave as a tiny magnet. In the absence of an applied field, these tiny magnets are randomly oriented, but when they are put in the presence of an applied magnetic field (ho), they will align with or against that field. A slight excess will align with the field ( -spin) while the others will be anti parallel ( -spin). (cid:149) 1h has a spin quantum number i = 1/2 and has allowed spin states of +1/2 or -1/2. 1 (cid:149) other nuclei with i = 1/2 are 13c, 19f and 31p and these also respond to an external magnetic field (cid:149) the nuclei of nmr-active nuclei behave like tiny bar magnets (cid:149) These concepts apply to 1h, 13c, and 19f nuclei which have spin quantum numbers of.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232