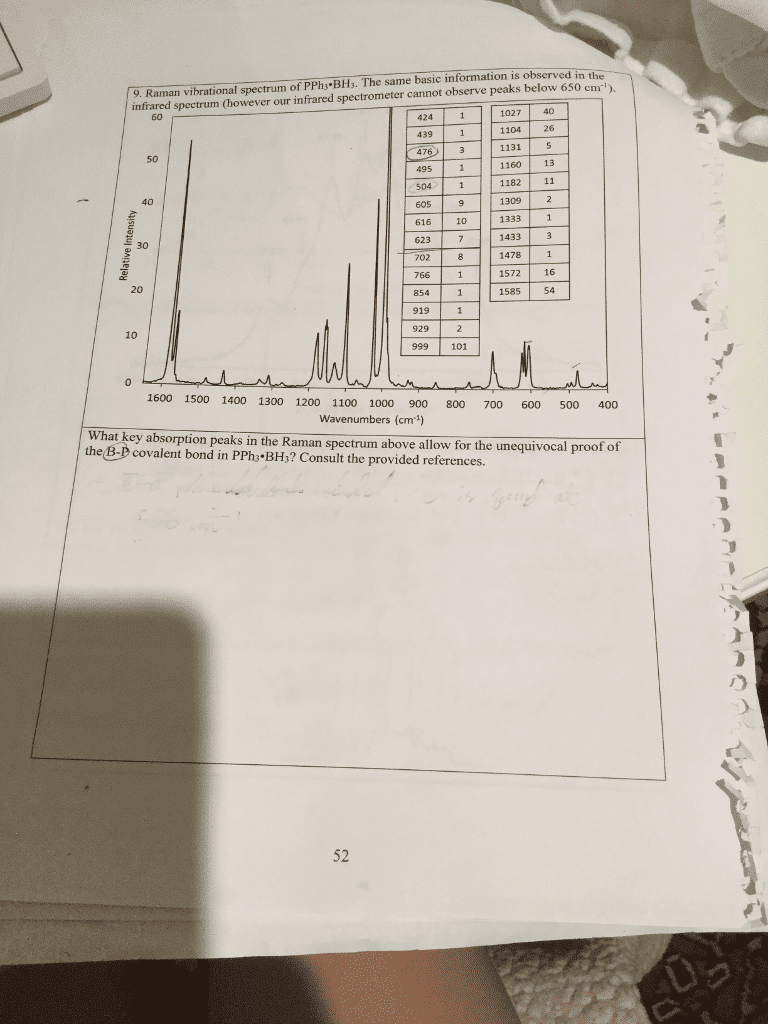
CHEM 1300 Lecture Notes - Lecture 18: Antibonding Molecular Orbital, Energy Density, Alanine
Document Summary
For all n, c and o: determine number of electron groups, determine the ideal bond angles, determine the hybridization, describe the bonding. Localized bonds do not explain color, conductivity and some molecular structures. Delocalized electrons are electrons shared between 3 or more atoms. Mos have been referred to briefly in class before, now they will be formally included. 1: pure s and p atomic orbitals of the atoms in a molecule combine to produce orbitals that are spread out, or delocalized, over several atoms, leading to molecular orbitals (mos), advantages of mos over localized bonding theory: Predicts relative bond lengths and energies a: predicts magnetic properties, correctly explains electronic structures of molecules which do not follow lewis. 1st principle: the total number of molecular orbitals produced by a set of interacting atomic orbitals is always equal to the number of interacting atomic orbitals. These orbitals can be added or subtracted.