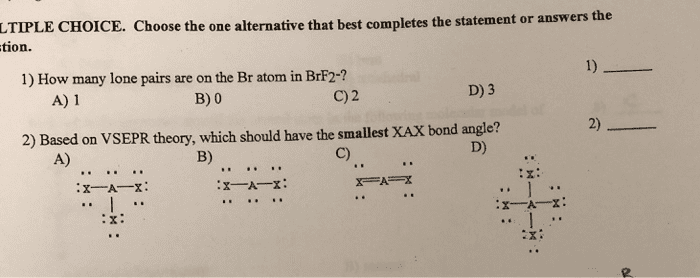CHEM 1300 Lecture Notes - Lecture 21: Hexane, Pentane, Electronic Band Structure
Document Summary
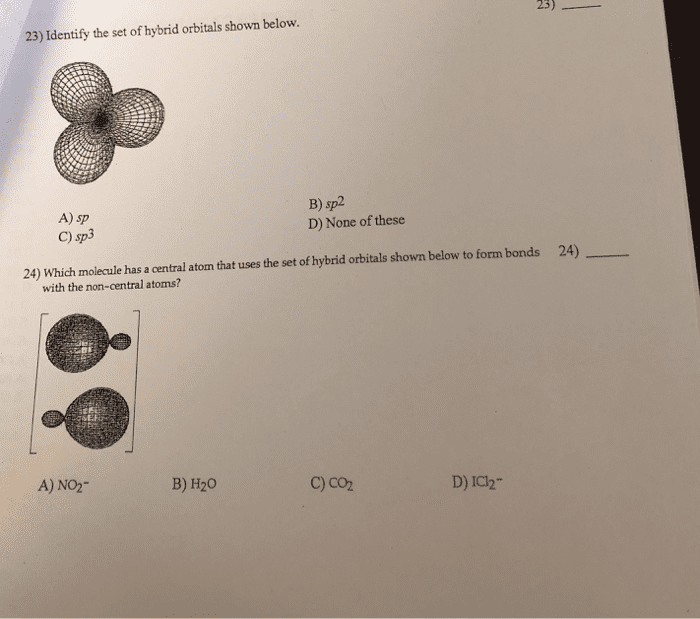
Review: how many antibonding molecular orbitals will form if six atomic orbitals interact, 2, 3, 5, 6, 8, what is the bond order of 2+ , 2. 5, 2, 1. 5, 1, 0. 5. E(cid:454)te(cid:374)ded overlappi(cid:374)g or(cid:271)itals are(cid:374)"t li(cid:373)ited to p-orbitals. Metal orbitals (s, p, d and f) can overlapping to form molecular orbitals. As the number of overlapping atomic orbitals increases, the number of molecular orbitals increases and the energy difference between the orbitals decreases. When there are hundreds or thousands of overlapping atomic orbitals, the resulting molecular orbitals are very (cid:272)lose i(cid:374) e(cid:374)erg(cid:455) so (cid:272)lose that the(cid:455) (cid:272)a(cid:374)"t (cid:271)e easil(cid:455) distinguished. When a potential is applied, electrons flow through the metal from the negative end to the positive end (through the orbitals). Band gap (eg) the energy difference between the filled bonding orbitals and the empty antibonding orbitals. Semiconductors compounds that become conductors in the presence of an appropriate energy source.