CHEM120 Lecture 11: c123-module1-w18
112 views62 pages
20 Mar 2018
School
Department
Course
Professor
Document Summary
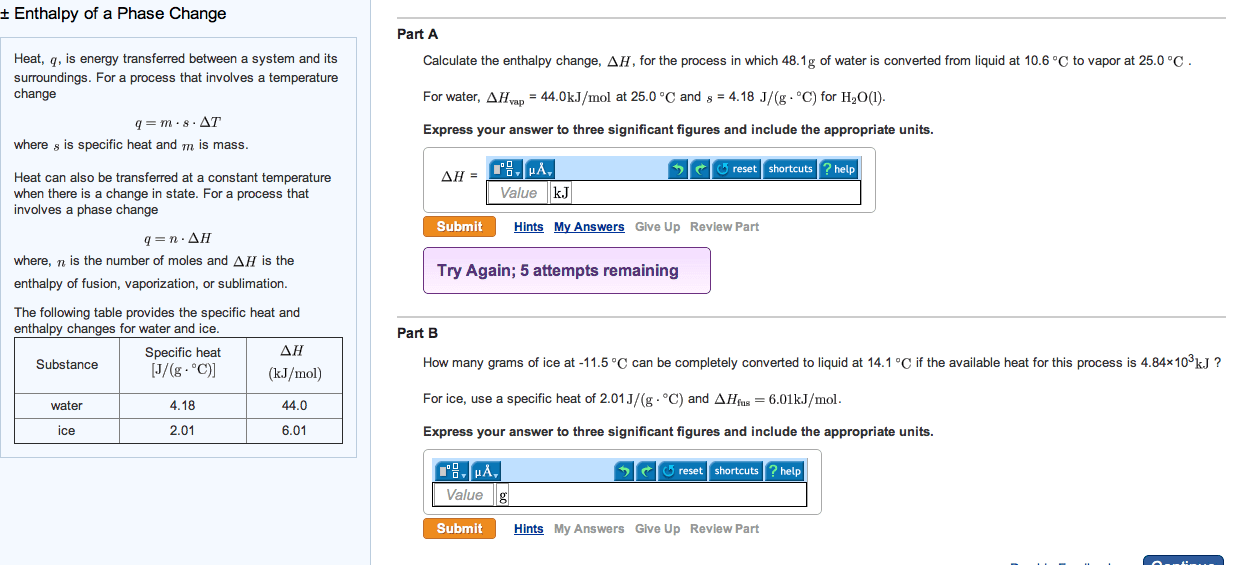
Module 1: energy, entropy, and spontaneous change page 1 of 62. In this module, we focus our attention on a system which undergoes some transformation between two states a and b: It may occur in one step or may involve many steps. In a physical change, there is no change in the chemical composition of the system. However, there may be changes in temperature, pressure, volume, density, or the phase (state of aggregation). Examples of physical transformations include heating, cooling, expansion, compression, and phase changes, such as vaporization, fusion (melting), and sublimation. In a chemical change, the chemical composition of the system is changed by a chemical reaction. Energy is conserved. (this is the first law of thermodynamics. ) Entropy increases. (this is the second law of thermodynamics. ) These statements illustrate that the concepts of energy and entropy are central to understanding or predicting physical and chemical changes.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232