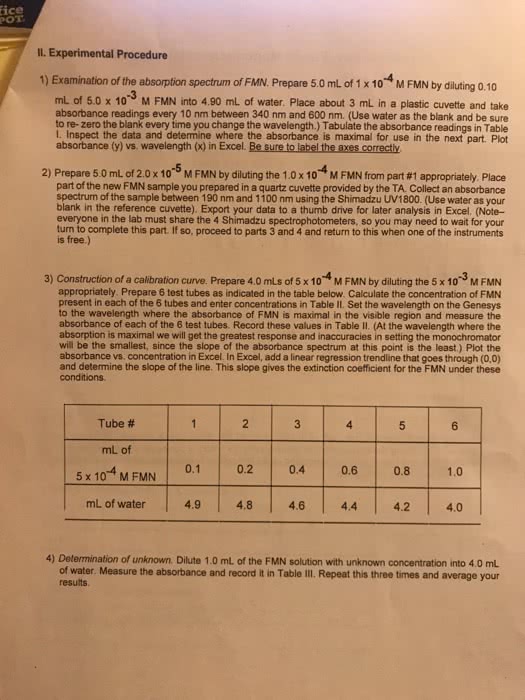
CHEM237L Lecture Notes - Diabetes Mellitus Type 1, Blood Sugar, Chromophore
304 views2 pages
15 Jul 2014
School
Department
Course
Professor
Document Summary
The absorptivity coefficient will be the same between the standards (s) and the unknowns (u) because we are using the same light absorbance species (glucose). Therefore, the absorbance equation will be a = l c. The path-length should also stay constant between the standards and the unknowns because we are using the same volume in each of the wells, therefore: l s = lu. Now we can set the standards and unknowns equal to each other and solve for the concentration of the unknown as follows: Q3: the absorptivity constant and the concentration are constant. The path length is dependent on volume; therefore we can solve this problem: The absorbance would be 0. 0989 if the volume was 220ul: they would determine the concentration to be this: Two possible interfering substances for this enzymatic method for determining the amount of glucose in your blood would be:
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232