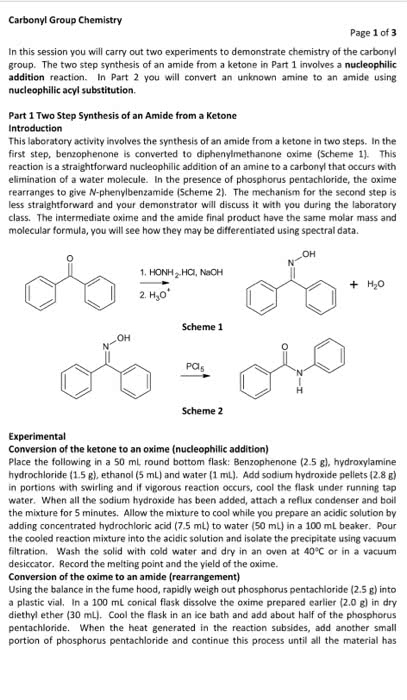
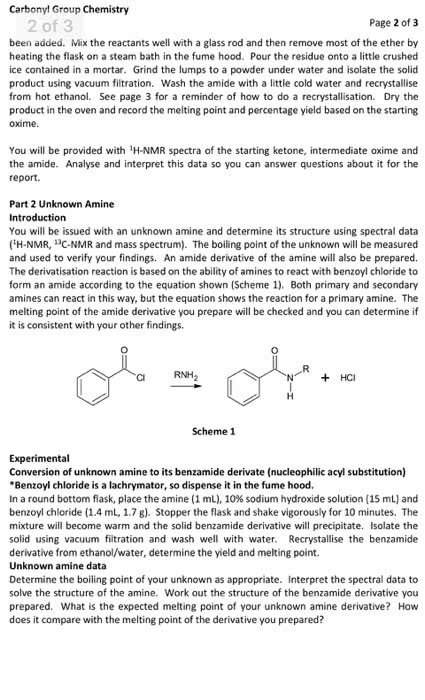
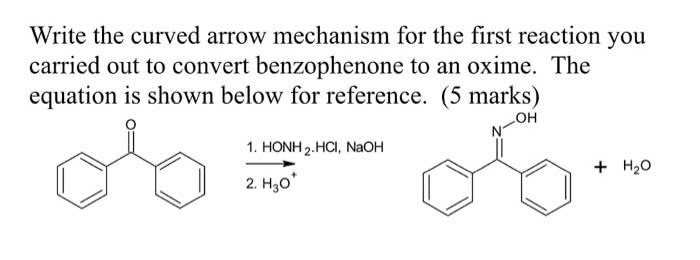
CHEM 121 Lecture Notes - Petri Dish, Purified Water, Litmus
330 views2 pages

79
CHEM 121 Full Course Notes
Verified Note
79 documents
Document Summary
The initial mass of aluminum is required to calculate the theoretical yield: add koh solution into a beaker over a hotplate containing the aluminum pieces under the fume hood. The hotplate will speed up the dissolution process. Stirring the beaker will also help the dissolution process. Do this under the fume hood because hydrogen gas is produced in the reaction: 2al(s) + 2koh(aq) + 6h2o(l) . 2al(oh)4- (aq) + 2k+aq) + 3h2(g): make sure that the solution is not over 30ml. If the solution is over 30ml, you can evaporate some of the water out using the hotplate (at a later step): remove any impurities from the solution by vacuum filtration. The filter flask should be extremely clean so that impurities do not alter the concentration or volume of your solution. Make sure you do this step while the solution is still hot so that the impurities don"t cool down and dissolve.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
Part 1: Separation of Iron filings
- Mass a watchglass and record
- Obtain a Kimwipe tissue from the top of the benches and a rodmagnet
- Wrap the Kimwipe tissue around the magnetic end, holding thetissue tight to the rod magnet.
- Swirl the Kimwipe in your mixture. Knock off excess sand beforemoving on to step 5
- Carefully over the watch glass remove the Kimwipe tissue fromthe magnet so the iron falls on the the watch glass
- Repeat steps 4 and 5 two more times, make sure you record howsuccessful this separation was � does the evaporating dish containonly iron?
- Mass the watchglass and the collected iron.
- Dispose of the iron in the hood.
Part 2: Separation of N
- Mass a clean, dry, evaporating dish.
- Pour the leftover mixture from step 1 into the evaporating dishand mass.
- Place the evaporating dish on a hot plate in the hood.
- Gently and slowly heat the mixture until the white fumes cease.Be patient. Depending on the amount of ammonium chloride in yoursample, this could take quite a while. A half our would not beunheard of.
- Stir the mixture frequently to facilitate the sublimation ofthe ammonium chloride. This is crucial in order to obtain goodresults. (Use crucible tongs to hold onto the dish so it doesn�tmove as much)
- After the white fumes disappear, turn off the hot plate andremove the evaporating dish with crucible tongs and let it cool inthe hood.
- When the evaporating dish is cooled, mass the left overmaterial and the evaporating dish.
Part 3: Determining the amount of NaCl.
- Measure 10 mL of distilled water in a graduated cylinder.Slowly add the water to the mixture in the evaporating dish whilecontinuously stirring with a clean, glass stirring rod. Afteradding all of the water, stir for 2 min more.
- Assemble the gravity filtration apparatus shown in Figure1.
- Fold a piece of filter paper as shown in Figure 2. Open up thepaper cone, and place it in the filtering funnel.
- Position a beaker under the funnel, making sure that the funnelstem is touching the inside wall of the beaker. Pour as much of thesupernatant liquid as possible from the evaporating dish intofunnel. Collect the filtrate in the beaker.
- Transfer the solid remaining in the evaporating dish into thefunnel, using a stream of distilled water from a wash bottle, asshown in Figure 3.
- Re mass the watchglass from Part 1 and record this mass.
- Pour the filtrate in the beaker into the evaporating dish
- Place the evaporating dish containing the filtrate on a hotplate. Begin heating the solution.
- While the solution is heating to boiling, move onto Part 4
- Carefully remove the filter paper and residue from the funnel.Place the paper and residue onto the watchglass.
- Place the watchglass with the filter paper and sand on top ofthe boiling salt solution to dry the sand
- Keep the salt water boiling, until there is just a little bitof water left, or popping sounds are heard, then turn off the hotplate, and allow the heat from the hot plate to finish evaporatingand drying your salt.
- Allow the evaporating dish and its contents to cool to roomtemperature.
- Obtain another watch glass and mass.
- Transfer the dried sand and filter paper from original watchglass (caution, salt has spattered onto this watch glass, so don�tknock any off, that is product you will need) onto the new cleanmassed watch glass. Replace the original watch glass onto thecooling evaporation dish
- Mass the new watch glass, sand and filter paper all together.Discard filter paper and sand into garbage. Observe how pure yoursand looks.
- After evaporating dish has cooled, determine the mass of theevaporating dish, watch glass plus NaCl. Record this mass. Then,discard the NaCl as directed. Wash evaporating dish and watch glassto dissolve the salt.
- Observe how pure your salt looks.
Result and Discussion: Separation Lab
Substance | Grams recovered | % mass |
Fe | ||
NH4Cl | ||
NaCl | ||
SIO2 |
Overall percentRecovery:
A) Explain what steps(s) youcould have improved (and how) to help your percentrecovery.
b) Write a paragraph about thelab include briefly what was done, your result, were you correctand possible experimental errors- do not state human measurementerrors as errors. Be Specific, what specific human errors occurred,what measurement errors occurred, what process in the experimentcould errors have occurred.