CHEM 121 Lecture Notes - Lecture 10: Spherical Coordinate System, Principal Quantum Number, Angular Momentum
CHEM 121 verified notes
10/38View all
9
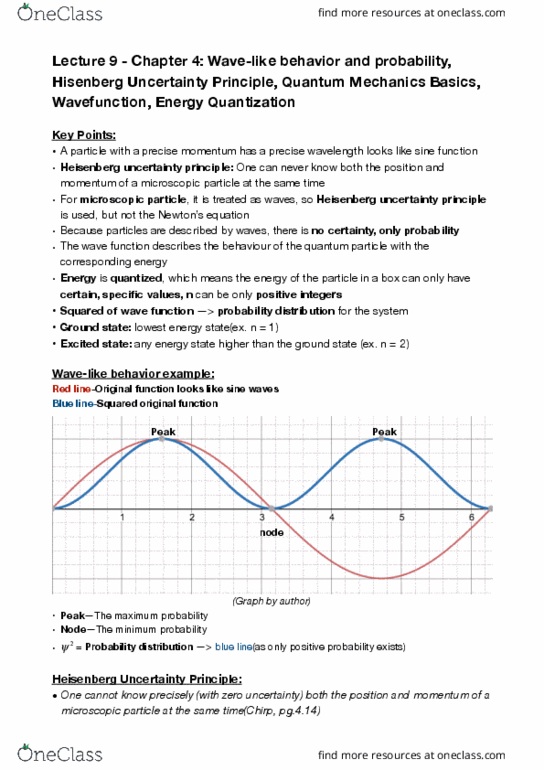
CHEM 121 Lecture Notes - Lecture 9: Uncertainty Principle, Probability Distribution, Potential Energy
10

CHEM 121 Lecture Notes - Lecture 10: Spherical Coordinate System, Principal Quantum Number, Angular Momentum
11
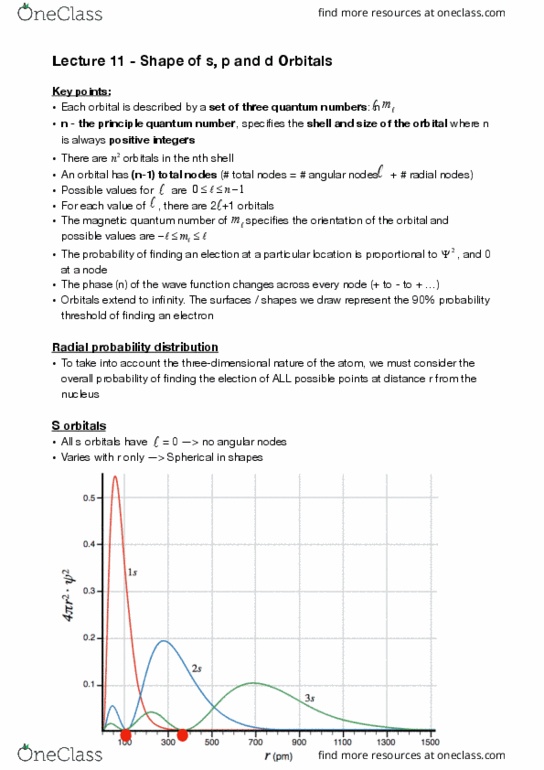
CHEM 121 Lecture Notes - Lecture 11: Principal Quantum Number, Magnetic Quantum Number, Exponential Decay
Document Summary
Lecture 10 - quantum mechanics exercises, chapter 5. Known values: l = 10 = 10 x 10-10 m = 1 x 10-9 m, m = mass of proton = 1. 67 x 10-27 m, n = 2 and n = 3, h = 6. 626 x 10-34 js. Unknown value: the energy di erence between n=3 and n=2. Known values: e = 1. 6 x 10-19 j, l = 30 = 3 x 10-9 m, m = mass of electron = 9. 11 x 10-31 kg, h = 6. 626 x 10-34 js. E1 (n2 12 )(6. 626 10 34 js)2. N2 = 25. 9 n = 4. 99 5. Chapter 5: electronic structure of atoms and ions. Key points: one electron atom or ion are called as atomic orbitals, distance between nucleus and electron is given by r, coulomb"s law: potential energy of attraction between two charges %




