CHEM 201 Lecture Notes - Lecture 8: Pre-Exponential Factor, Rate Equation, Reaction Rate Constant
73 views41 pages
Document Summary
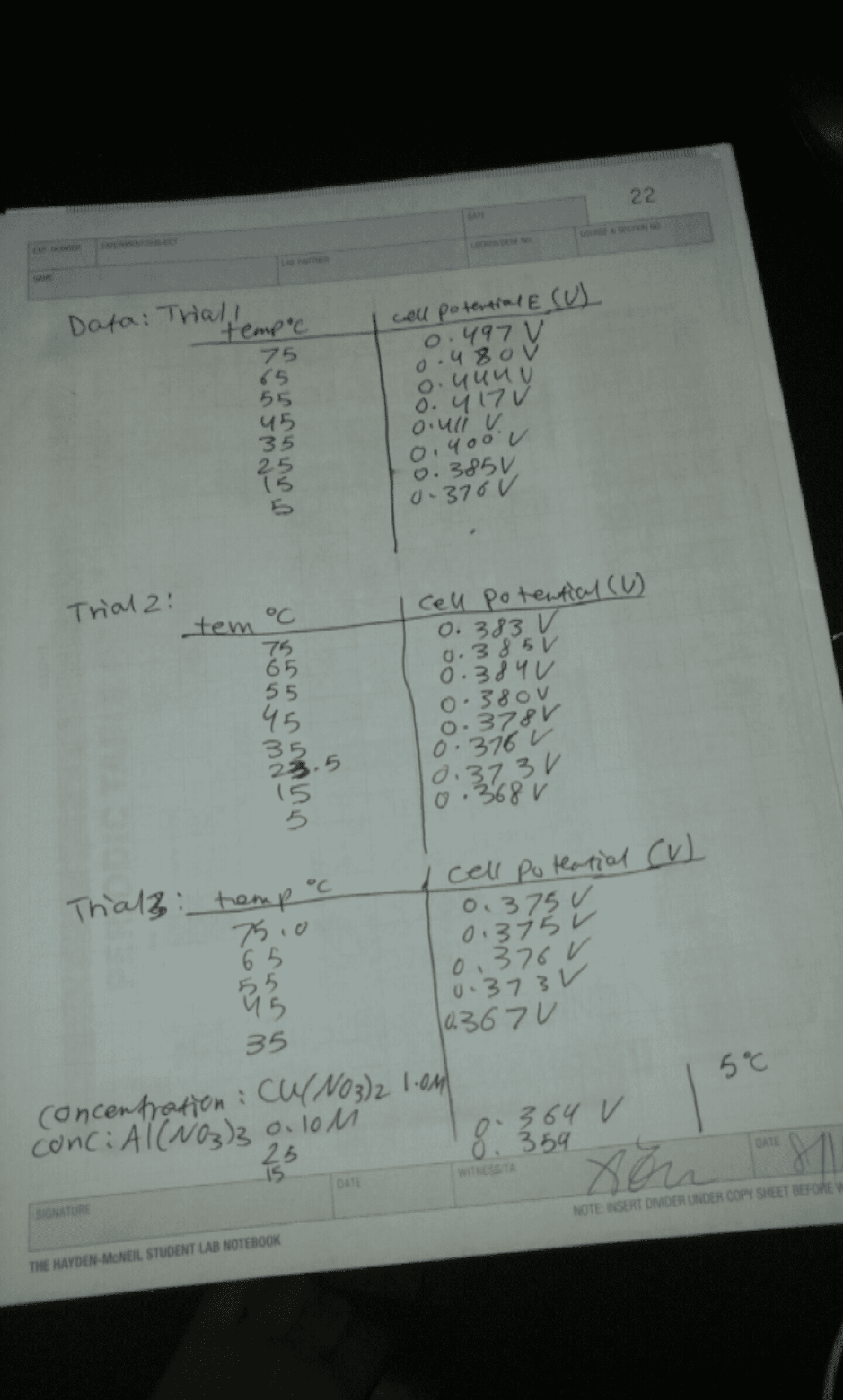
How to speed up or slow down a process. Experimentally measure concentration [a] (or any quantity [a], V (cid:1) rt for a gas) as a function of time t. such as pressure pa = (cid:0)na. Fig k1 get equil value [a]eq at t = . De ne rate or velocity v of reaction (a +ve quantity) by slope at t rate v = d[a] dt could also measure [b] or [c]: note stoichiometry is important v = d[a] dt. In general, no way to predict rates of chemical reactions - must be determined experimentally. Many organic and inorganic reactions take minutes to hours. Experiment involves measuring concentration (or a related quantity) as function of time. For faster reactions: volume of gas, change in optical rotation, change in colour intensity (beer"s law, change in total p = pi pi at constant v for gas-phase reaction (must then relate p to p (one species, etc.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232