CHEM1001 Lecture 5: Chem 5
Document Summary
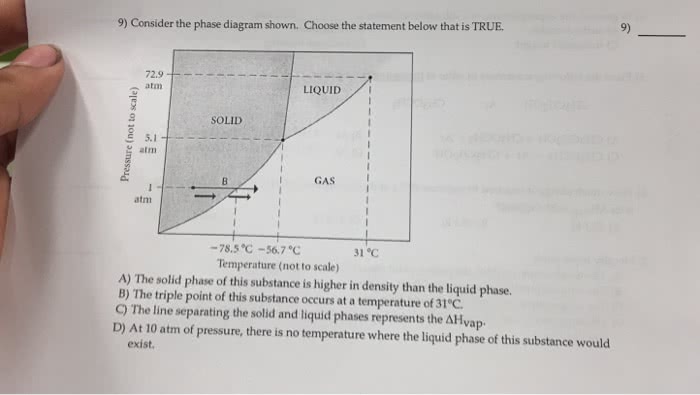
When a solid is heated above its melting point, it becomes liquid because the pressure is higher than the triple point of the substance. Intermolecular forces are still important, but the molecules have enough energy to move around, which makes the structure mobile. This means that a liquid is not definite in shape but rather conforms to the shape of its container. Its volume is usually greater than that of its corresponding solid. The highest temperature at which a particular liquid can exist is called its critical temperature. A liquid can be converted to a gas through heating at constant pressure to the substance"s boiling point or through reduction of pressure at constant temperature. This process of a liquid changing to a gas is called evaporation. Gas molecules have either very weak bonds or no bonds at all, so they can move freely and quickly.