CHEM1001 Lecture Notes - Lecture 16: Measuring Cup, Laboratory Glassware, Graduated Cylinder
56 views2 pages
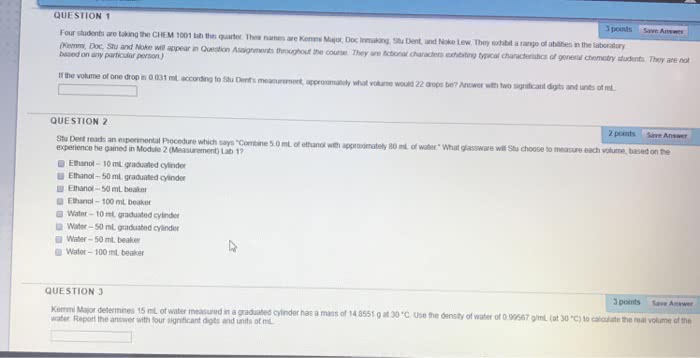
Document Summary
The properties of a material may be described in many ways. If you have two containers of water that are different sizes, they each hold a different amount, or volume, of water. The unit for volume is a unit derived from the si unit of length and is not a fundamental si measurement. If two water samples have different volumes, they still share a common measurement: the density. Density is another measurement derived from si basic units. The density of a material is defined as its mass per unit volume. Each volume of water is different and therefore has a specific and unique mass. The mass of water is expressed in grams or kilograms, and the volume is measured in liters, cubic centimeters, or milliliters. Density is calculated by the dividing the mass by the volume, so that density is measured as units of mass/volume, often g/ml.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232