CHEM1006 Lecture Notes - Lecture 2: Dimensional Analysis
Document Summary
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
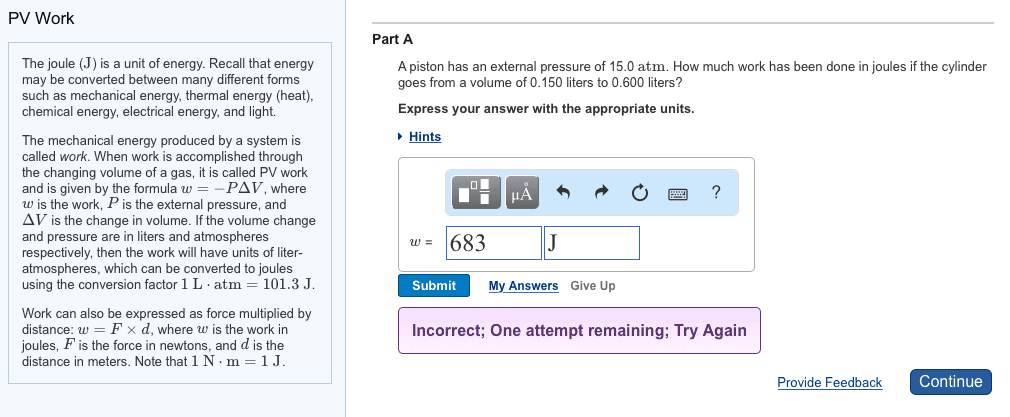
| PV Work The joule (J) is a unit of energy. Recall that energy may be converted between many different forms such as mechanical energy, thermal energy (heat), chemical energy, electrical energy, and light.The mechanical energy produced by a system is called work. When work is accomplished through the changing volume of a gas, it is called PV work and is given by the formula w=âPÎV, where w is the work, P is the external pressure, and ÎV is the change in volume. If the volume change and pressure are in liters and atmospheres respectively, then the work will have units of liter-atmospheres, which can be converted to joules using the conversion factor 1 Lâ atm=101.3 J. Work can also be expressed as force multiplied by distance: w=FÃd, where w is the work in joules, F is the force in newtons, and d is the distance in meters. Note that 1 Nâ m=1 J. | Part A A piston has an external pressure of 9.00 atm. How much work has been done in joules if the cylinder goes from a volume of 0.160 liters to 0.550 liters? Express your answer with the appropriate units. Hints
|
To understand the relationship between the equilibrium constant and rate constants.
For a general chemical equation
A+BâC+D
the equilibrium constant can be expressed as a ratio of the concentrations:
Kc=[C][D][A][B]
If this is an elementary chemical reaction, then there is a single forward rate and a single reverse rate for this reaction, which can be written as follows:
forward ratereverse rate==kf[A][B]kr[C][D]
where kf and kr are the forward and reverse rate constants, respectively. When equilibrium is reached, the forward and reverse rates are equal:
kf[A][B]=kr[C][D]
Thus, the rate constants are related to the equilibrium constant in the following manner:
Kc=kfkr=[C][D][A][B]
Part A
For a certain reaction, Kc = 5.79Ã108 and kf= 634 Mâ2â sâ1 . Calculate the value of the reverse rate constant, kr, given that the reverse reaction is of the same molecularity as the forward reaction.
Express your answer with the appropriate units. Include explicit multiplication within units, for example to enter Mâ2â sâ1include â (multiplication dot) between each measurement.
| | |||
| kr = |
SubmitHintsMy AnswersGive UpReview Part
Part B
For a different reaction, Kc = 7.92Ã105, kf=6.77Ã105sâ1, and kr= 0.855 sâ1 . Adding a catalyst increases the forward rate constant to 2.24Ã108 sâ1 . What is the new value of the reverse reaction constant, kr, after adding catalyst?
Express your answer with the appropriate units. Include explicit multiplication within units, for example to enter Mâ2â sâ1include â (multiplication dot) between each measurement.
| | |||
| kr = |
SubmitHintsMy AnswersGive UpReview Part
Part C
Yet another reaction has an equilibrium constant Kc=4.32Ã105 at 25 âC. It is an exothermic reaction, giving off quite a bit of heat while the reaction proceeds. If the temperature is raised to 200 âC , what will happen to the equilibrium constant?
The equilibrium constant will
Yet another reaction has an equilibrium constant at 25 . It is an exothermic reaction, giving off quite a bit of heat while the reaction proceeds. If the temperature is raised to 200 , what will happen to the equilibrium constant?
| increase. |
| decrease. |
| not change. |