Any help/explanations with this Le Chatelier's Principle lab would be extremely helpful. I attemtped to do question A, but I'm not exactly sure if my answer is interpreted correctly. I have included the actual lab procedure with the 5 questions. Thank you again.





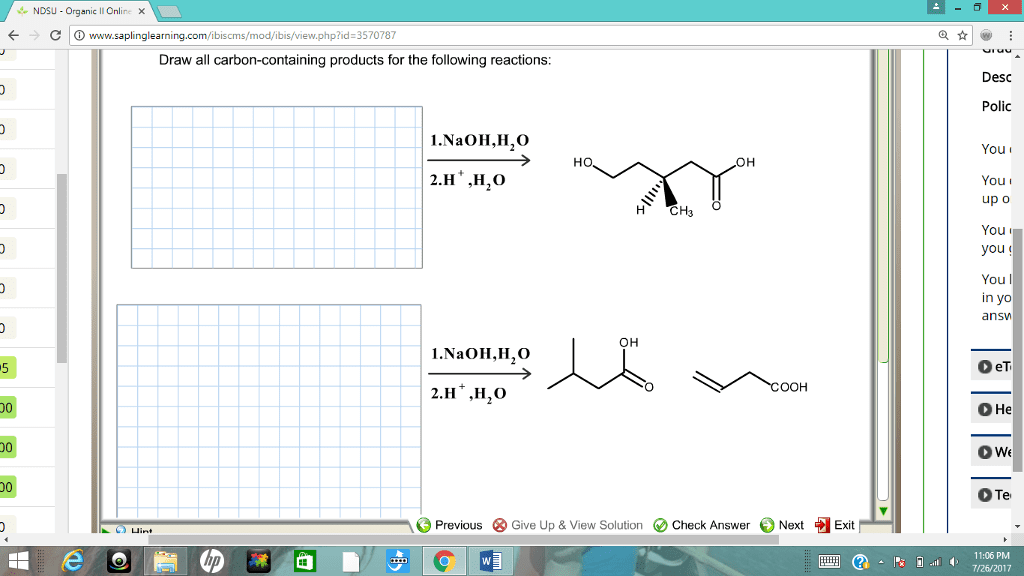
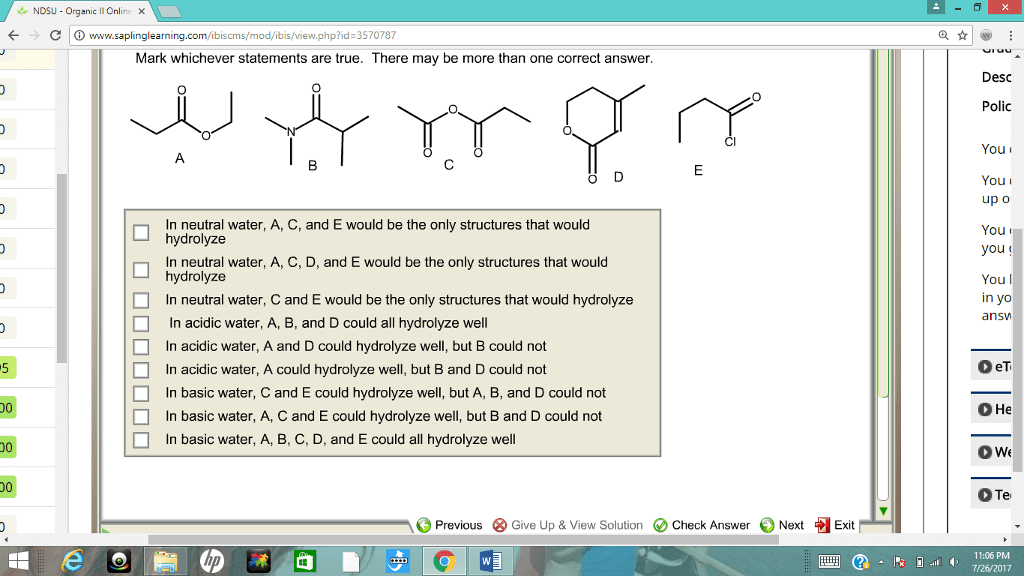
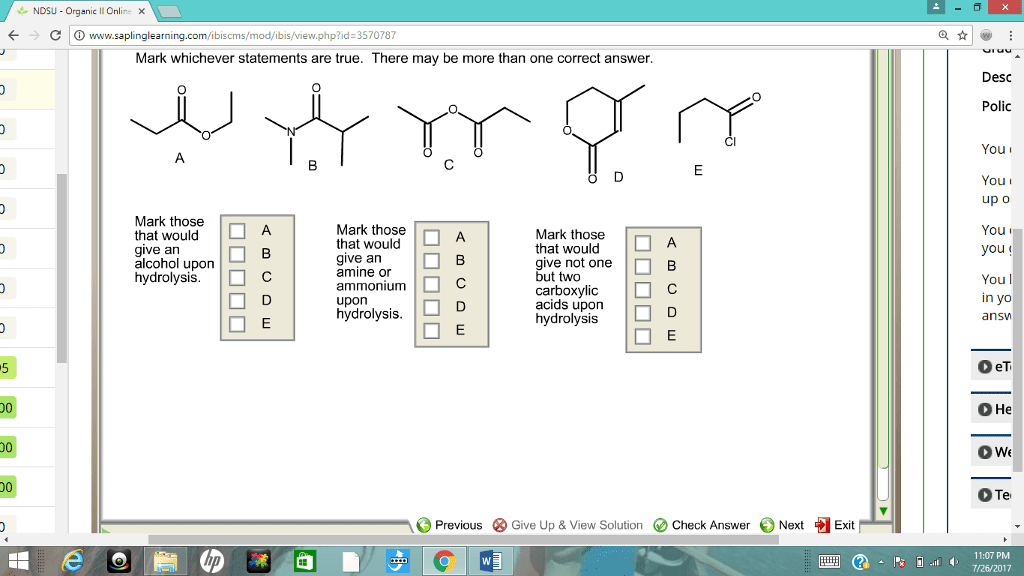
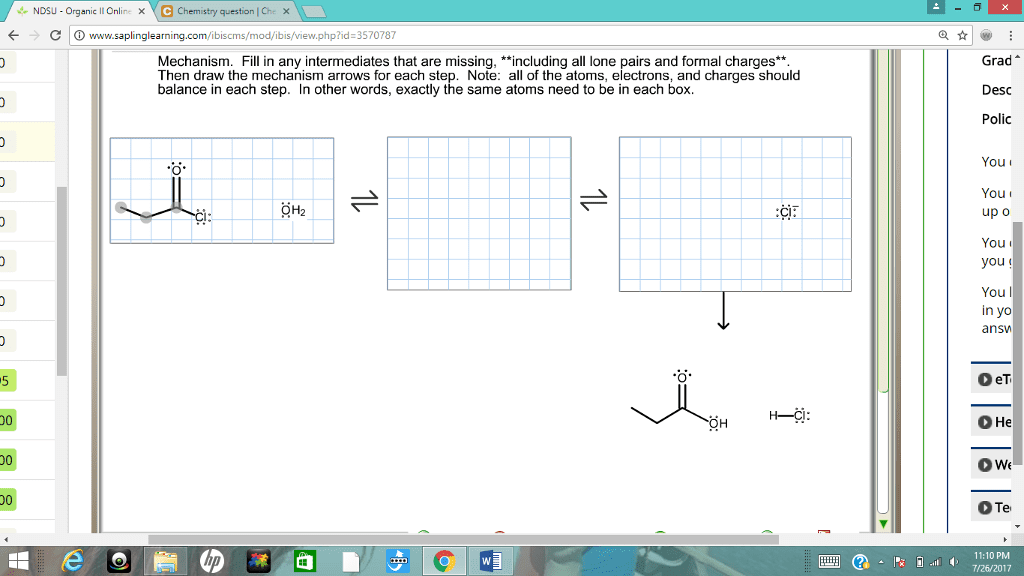
CHM 112 Le Chatelier's Principle Aim: To study how equilibrium systems respond when changes are introduced into them. Discussion: At equilibrium, the concentration of all the species remains constant. However, the forward and reverse reactions continue to occur. This is a dynamic system with no net change in concentration of reactants or products. Equilibrium systems are often disturbed or stressed by making changes in reaction parameters such as temperature, pressure or concentrations of reactants or products When these disturbances occur, the system responds in such a way that will counteract the stress. The Le Chatelier principle generalizes the behavior of equilibrium systems âwhen a stress is applied to a system in equilibrium, the system will respond (or shift) in such a way as to relieve the stress and restore equilibrium under a new set of conditions" Procedure: Solid, unionized methyl violet (HMV) is yellow, but it dissolves in water or alcohol to produce the violet anion (MV') according to the equation A. HMV (aq) H+ (aq) MV' (aq) ââ 1. Add about 5mL of distilled water to a regular test tube. Add a few drops of mehtyl violet indicator. Record the color of the solution. 2. Add 6M HCl drop by drop until a change in color occurs. Record. 3. Add 6M NaOH drop by drop until change in color occurs. Record 4. Explain your results in terms of Le Chatelier Principle B. Yellow Chromate ion (CrO42) can be converted to orange dichromate ion (Cr2O,') by the addition of sulfuric acid to a solution of potassium chromate 2CrO42-(aq) + 2H+è¹CnO72. (aq) + H2O 1. Mix 5ml of 0.1M K2Cr04 and 5 drops of IM H2S04. Observe and record results 2. Add 1M NaOH dropwise and note the color change 3. Add 1M H2S04 and note the color change. 4. Explain your results in terms of Le Chatelier Principle