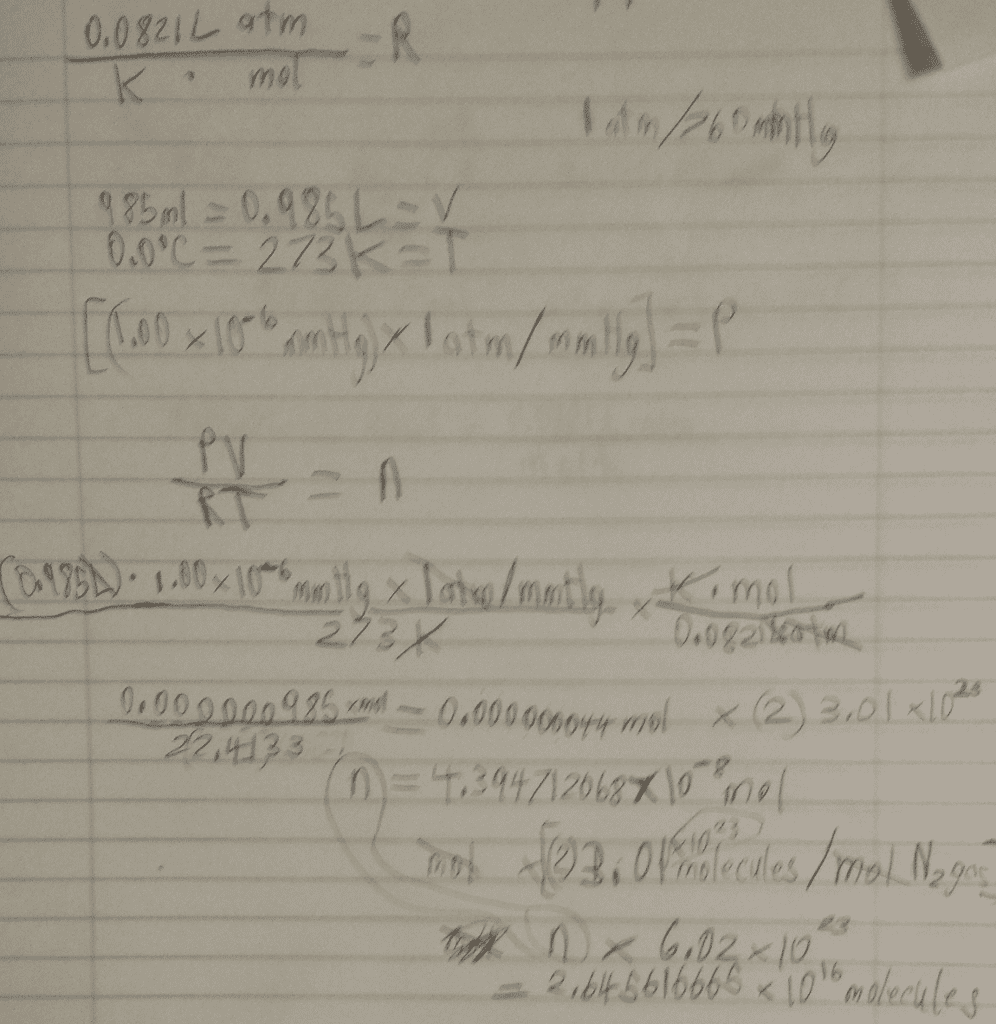CHM110H5 Lecture Notes - Lecture 9: Molar Volume, Horse Length, Ideal Gas
Document Summary
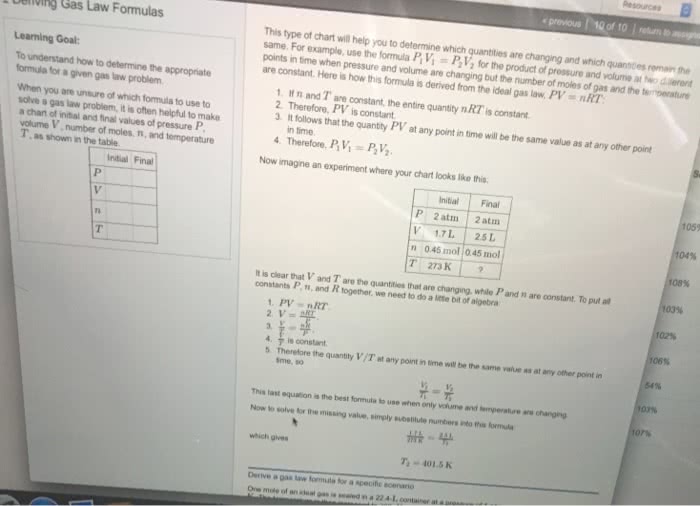
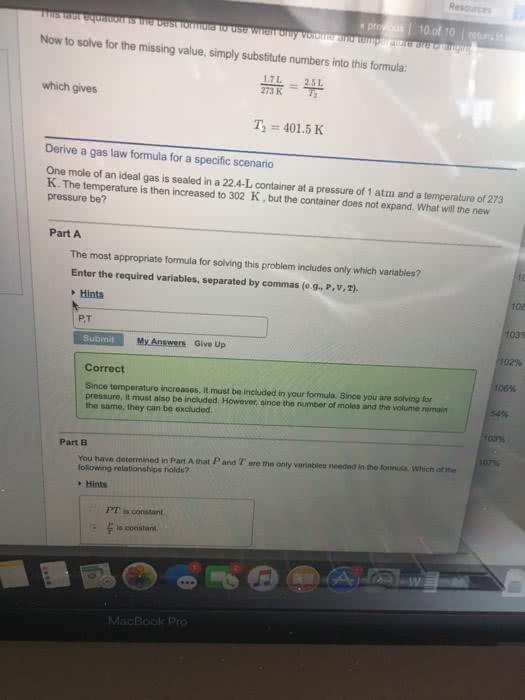
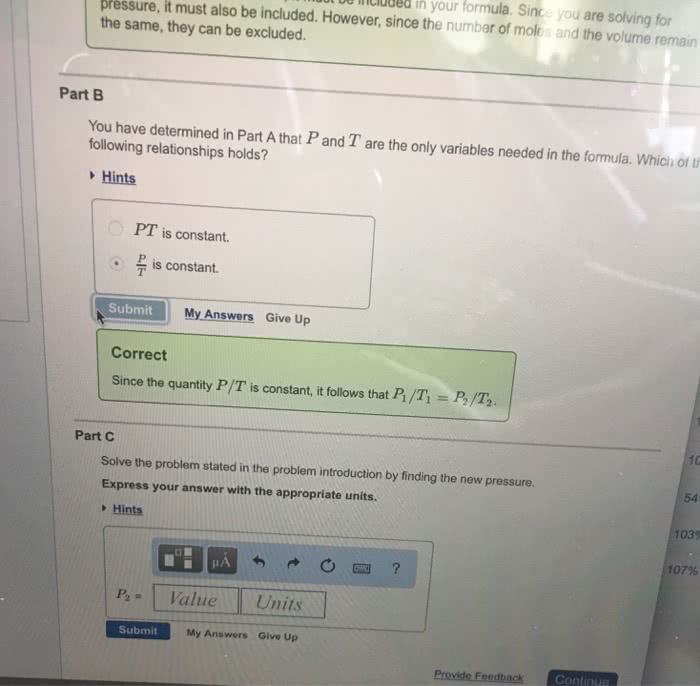
Lecture 9: relationship between v and n, gay-lussac"s law of. V n at constant t and p. Equal volume of gases under the same condition of temperature and pressure contains the same number of molecules. Gas constant r = pv/nt n t v p. R = 0. 08205 l atm / k mol. = 1. 987 cal / k mol (1 cal = One mole of an ideal gas occupies a volume of 22. 414. From this we can calculate the number of moles or molecules in any volume of gas at stp. For eg. how many moles are there in 10 l of a gas at. How many molecules are there in 10 l. [ remember one mole contains 6. 023 x 1023 molecules] Molar volume mol of any gas occupies 22. 4 l at stp. Assuming ideal behaviour predict which one will contain more number of molecules 2. 2. Which will have a greater volume at stp.