CHM120H5 Lecture Notes - Freezing-Point Depression, Molality
379 views1 pages
Document Summary
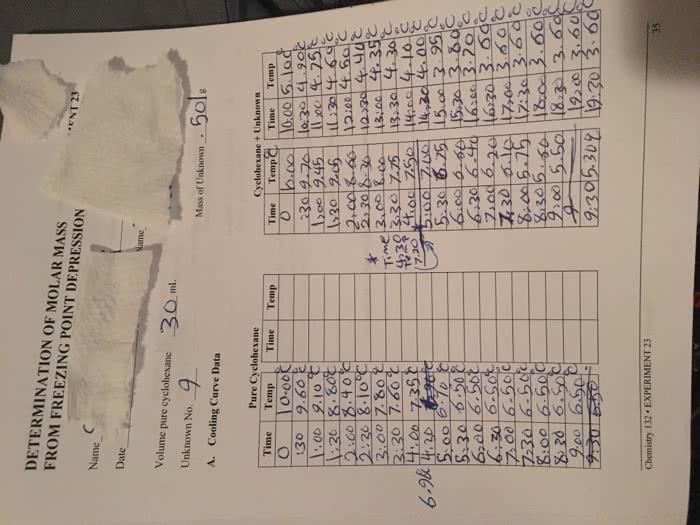
30 january, 2013: the temperature gradually drops as the solution is freezing because the concentration of the solution increases as the pure solvent in the solution freezes. This is because, in this experiment, we are interested in finding the change in temperature and not static values for temperature. As the temperature changes, the moles of solute to the volume of the solvent ratio changes which will eventually affect the molarity value despite having the same amount of solute in the solution. However, molality is measured in terms of mass of solute to the mass of solvent, which in this case is constant regardless of the temperature. Thus, molality is the unit used in measurements involving changes in temperature: using cyclohexane as a solvent is ideal for this experiment because it has a higher freezing point and freezing point depression constant.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers