Please take your time and find the correct answers to these questions and show all work and please explain so i can learn well. Thanks :-)
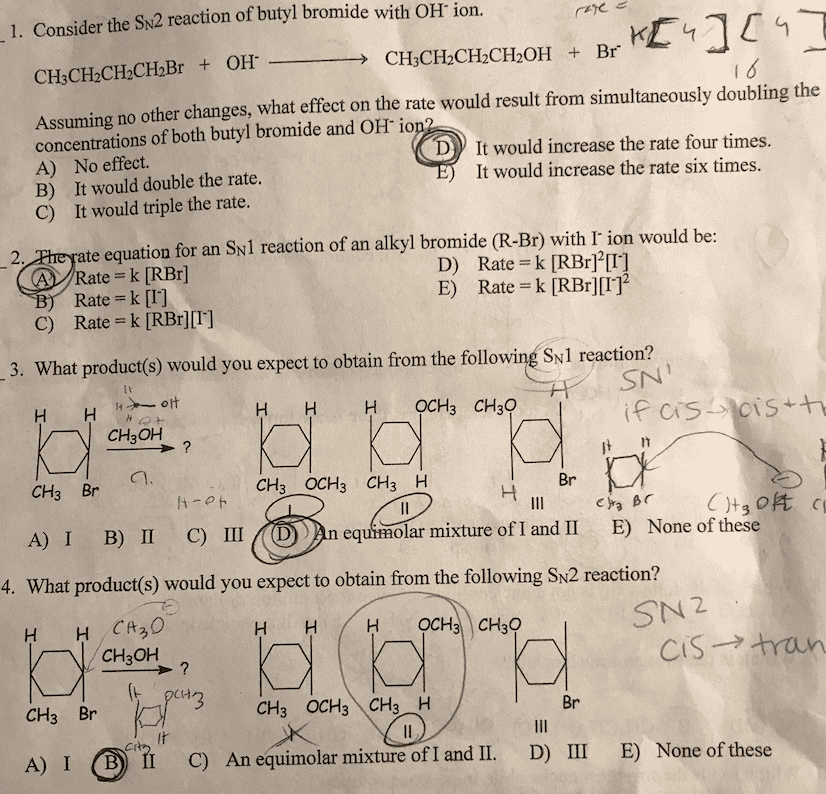
1. Consider the SN2 reaction of butyl bromide with OH ion. CHCH-CH.CHOH K Assuming no other changes, what effect on the rate would result from simultaneously doubling the concentrations of both butyl bromide and OH io D A) B) C) No effect. It would double the rate. It would triple the rate. It would increase the rate four times. It would increase the rate six times. te equation for an Sv1 reaction of an alkyl bromide (R-Br) with r ion would be ate k [RBr] 2 Rate = k [1] C) Rate k [RBr][I] D) Rate = k [RBr]2[1] E) Rate k [RBr][T] 3. What product(s) would you expect to obtain from the following Sy1 reaction? SN HOCH CHaO CH3OH CH3 Br CH3-OCH3 CH3 H Br A) I B) II IDn equhmolar mixture 4. What product(s) would you expect to obtain from the following SN2 reaction? IDAn equhmolar mixture of I and II E) None of these H OCH CH30 SN 2 CH3OH cs--> tran t-t CH3 Br CH3 OCH3 CH3 H Br C) An equim Olar mixtureofl and 11. D) III E) None of these