CHM120H5 Lecture Notes - Lecture 3: Freezing-Point Depression, Molar Mass, Colligative Properties
Document Summary
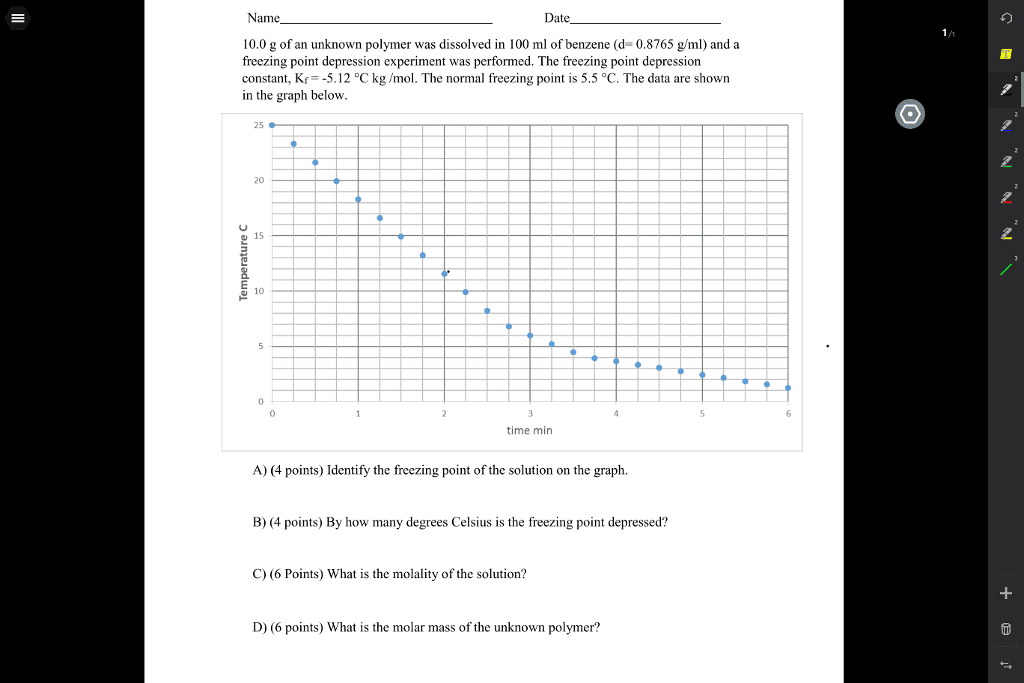
Determination of a molar mass by freezing point depression. The molar mass is one of the first properties that are determined for an unknown compound. One technique of determining molar mass is based on a colligative property of solutions, the freezing point depression. A colligative property is one that depends on the number of molecules in a solution and not on the nature of the molecules. Dissolving a non-volatile solute in a solvent results in a lowering of the equilibrium vapour pressure of the pure solvent. Consequently, the freezing point temperature of the solution is lower than that of the pure solvent. Freezing point depression has many practical applications including the use of salt to lower the freezing point of snow on the ground. The degree to which the freezing point is lowered depends upon the concentration of the solution (molality) , and the molal freezing point depression constant, kfp (characteristic of the solvent), as follows: