CHMA10H3 Lecture Notes - Lecture 3: Molar Mass, Limiting Reagent, Reagent
208 views3 pages
Verified Note
10 Sep 2018
School
Department
Course
CHMA10H3 verified notes
3/39View all
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related Documents
Related Questions
Why must chemical equations be balanced?
Choose all that apply.
Choose all that apply.
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict whether the given reaction is possible or not. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict whether the reaction requires a catalyst or not. | |
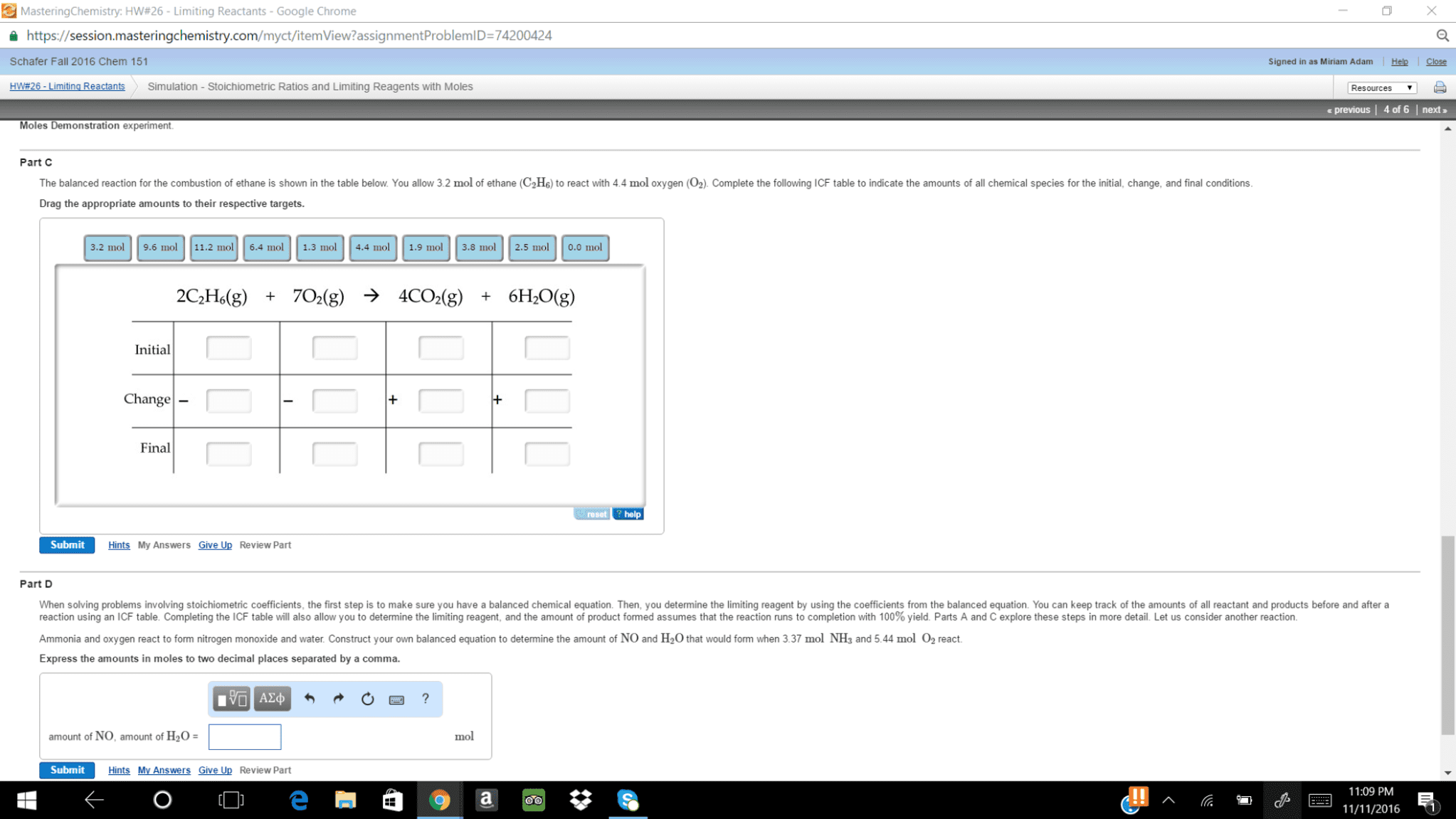
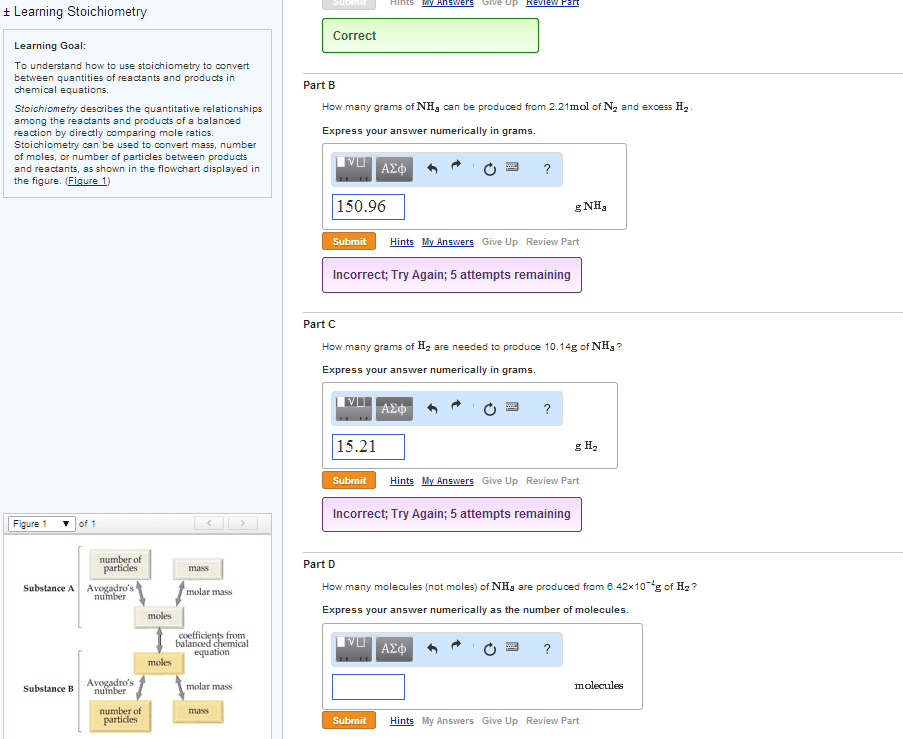
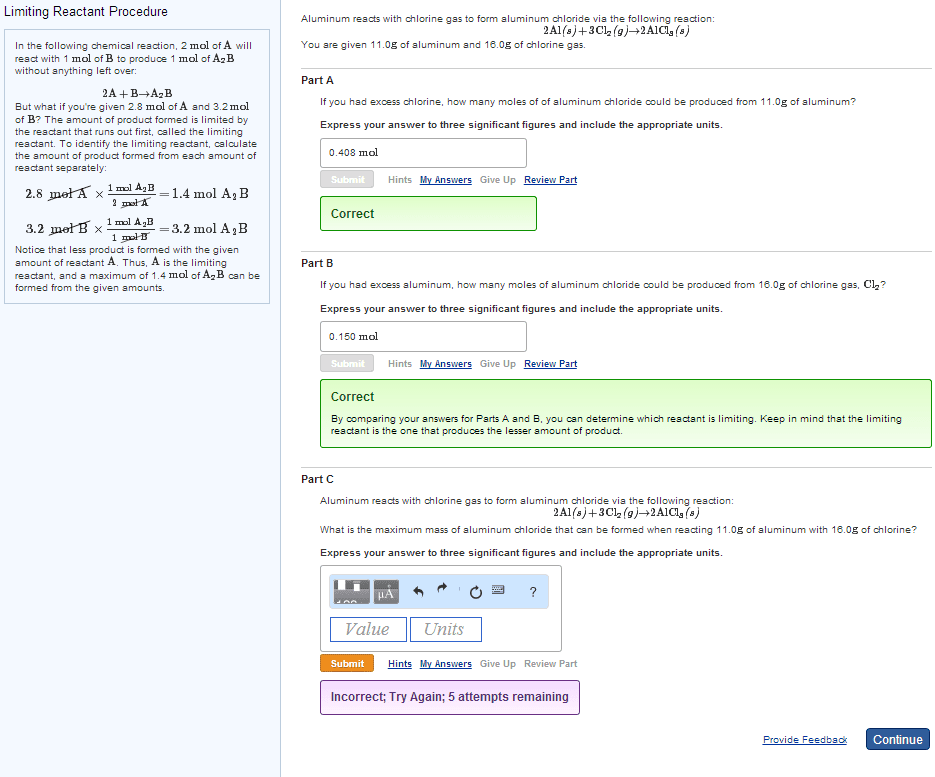
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict the amount of product that can form from a given amount of reactant. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict how much of one reactant is required to react with a given amount of another. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict how much reactants are required to form a given amount of products. |