CHMA10H3 Lecture Notes - Lecture 21: Freezing-Point Depression, Gas Constant, Formula Unit
CHMA10H3 verified notes
21/39View all
Document Summary
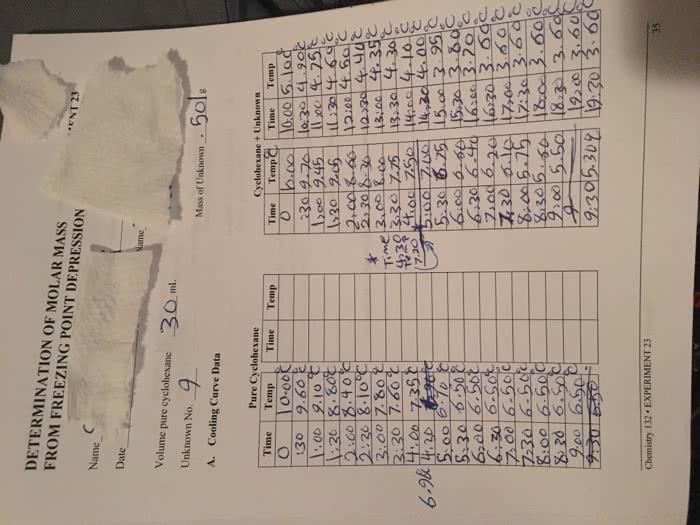
Freezing point depression: the freezing point of a solution is lower than the freezing point of a pure solvent when a non-volatile solvent is added to it: fpsolvent - fpsolution = tf = (m)(kf) Tf = change in the temperature of the freezing point of a solution compared to the freezing point of the pure solvent. Is usually a positive value: m = molality of solution, moles of solute/kg of solvent, kf = freezing point depression constant for the respective solvent used to make the solution. Boiling point elevation: the boiling point of a solution is higher than the boiling point of a pure solvent when a non-volatile solute is added: bpsolution - bpsolvent = tb = (m)(kb) Theoretical van"t hoff factor (cid:894)i(cid:895): ratio of the moles of solute particles to the moles of the formula units that are dissolved.