CHMA10H3 Lecture Notes - Lecture 22: Electron Configuration, Paramagnetism, Diamagnetism

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
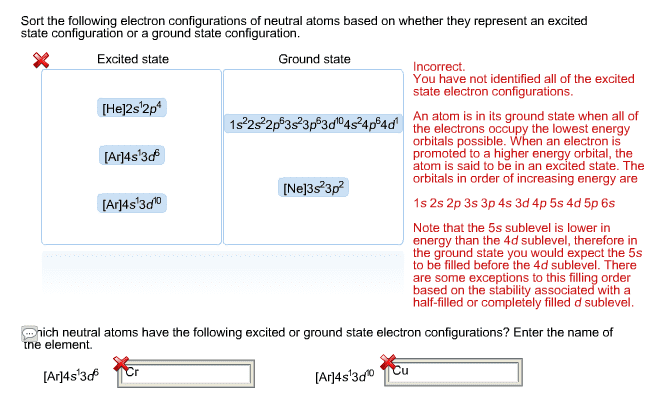
Chma10h3 introductory chemistry i: structure and bonding lecture 22: attention to. 8. 4 quantum mechanical model of electrons in atoms: summary of quantum numbers (n, l and m) and atomic orbitals, spin quantum numbers (ms) Diamagnetic and paramagnetic substances: orbital shape (yr2) S orbital (1s, 2s, 3s ) r90 indicates the radius of a sphere containing 90% of the electron density. P orbital (2p, 3p ): 3 perpendicular orbitals, 1 nodal plane. D orbital (3d ): 5 orbitals, 2 nodal planes. 8. 5 electron configurations in atoms (distribution of e in orbitals): aufbau principle: the lowest energy orbitals first, pauli exclusion principle, hund"s rule: occupy empty orbital before pair electrons. Z* = z s: s measure of the effect due to shielding. Slater"s rules for z*: shielding due to each of the other valence electrons = 0. 35, shielding due to each electron in the (n-1) shell = 0. 85, shielding due to each core electron = 1. 0 (n-2, n-3)