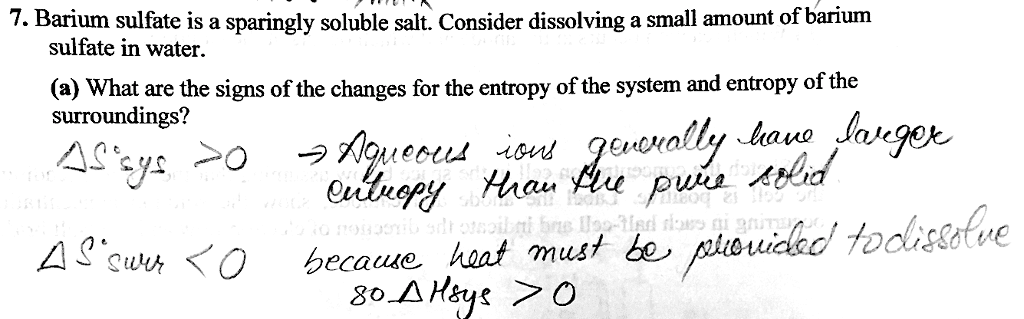CHMA11H3 Lecture 20: Chapter 17

36
CHMA11H3 Full Course Notes
Verified Note
36 documents
Document Summary
17. 3 - heat transfer and changes in the entropy of the surroundings: Heat transfer and changes in the entropy of the surroundings: A process that releases heat to the surrounding increases the entropy of the surroundings. A process that absorbs heat from the surrounding decreases the entropy of the. The magnitude of change in entropy of the surrounding is proportional to the magnitude: the microstates increased, qsystem is negative. ( ssurrounding) surroundings ( ssurrounding, ssurrounding -qsystem of qsystem, the microstates have decreased, qsystem is positive. The temperature dependence of ssurrounding: to the temperature. Change in energy of the surrounding ( ssurrounding) is inversely proportional. Freezing is a spontaneous process at low temperatures. The positive value of the change in energy of the surroundings and the negative value of the change in energy of the system give a positive net value for the change in energy of the universe. It is like giving dollars to a poor man.