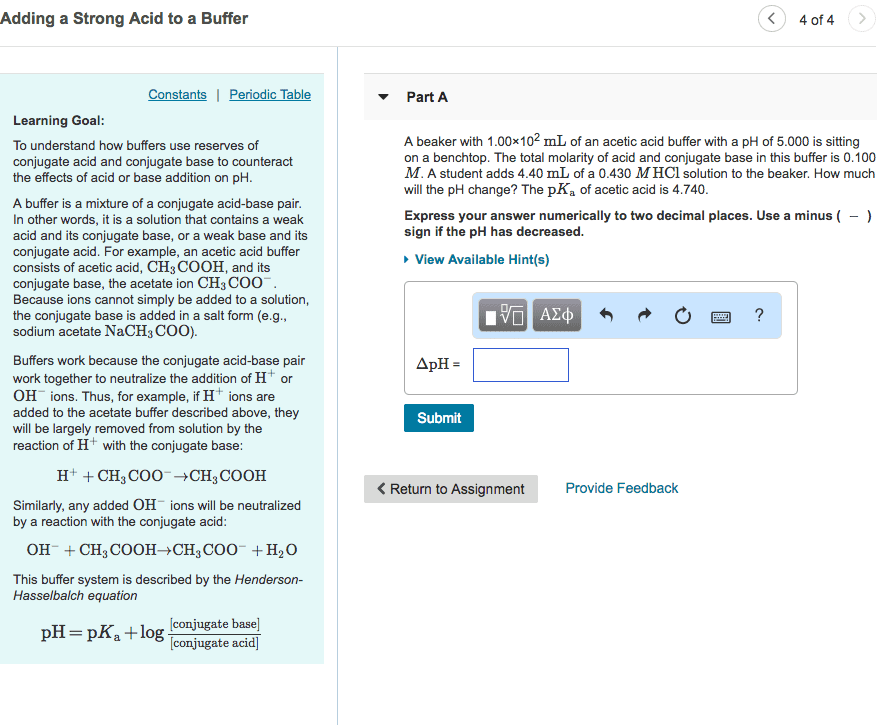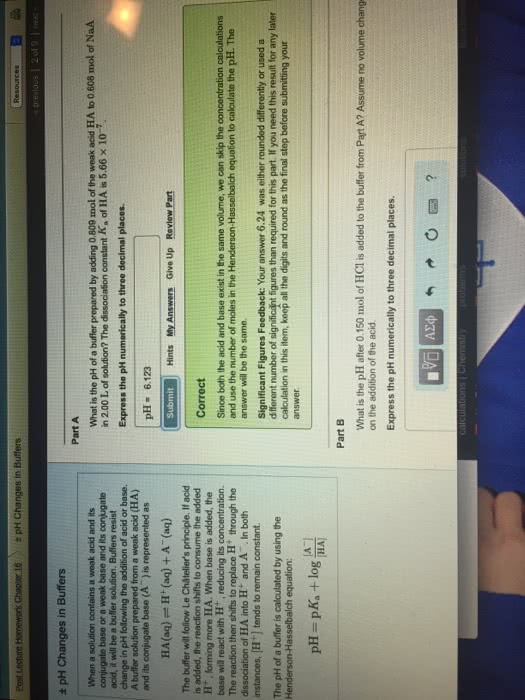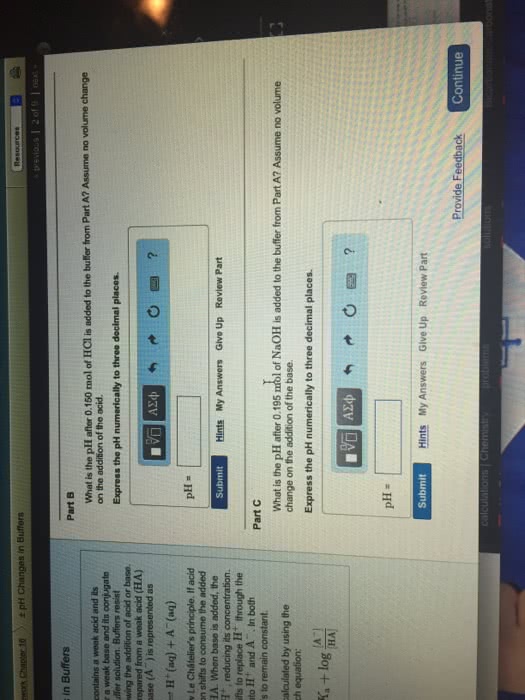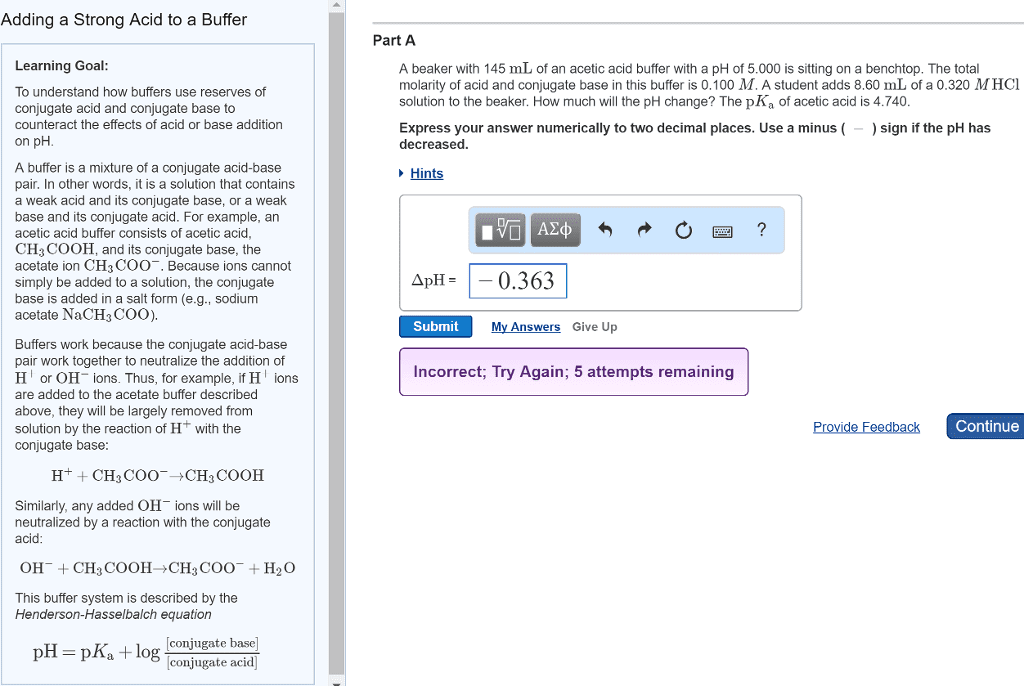
Adding a strong Acid to a Buffer Part A Learning Goal : A beaker with 145 mL of an acetic acid buffer with a pH of 5,000 is sitting on a benchtop. The total molarity of acid and conjugate base in this buffer is 0.100 M. A student adds 8.60 mL of a 0.320 M HC1 solution to the beakerHow much will the pH change? The pK, of acetic acid is 4.740. To understand how buffers use reserves of conjugate acid and conjugate base to counteract the effects of acid or base addition on pH Express your answer numerically to two decimal places. Use a minus (-) sign if the pH has decreased. Hints A buffer is a mixture of a conjugate acid-base pair. In other words, it is a solution that contains a weak acid and its conjugate base, or a weak base and its conjugate acid. For example, an acetic acid buffer consists of acetic acid, CH, COOH, and its conjugate base, the acetate ion CH3 COO. Because ions cannot simply be added to a solution, the conjugate base is added in a salt form (eg., sodium acetate NaCH COO). IVO Aze s o o o ? ApH -- 0.363 Submit My Answers Give Up Incorrect, Try Again; 5 attempts remaining Buffers work because the conjugate acid-base pair work together to neutralize the addition of H' or OH ions. Thus, for example, if H ions are added to the acetate buffer described above, they will be largely removed from solution by the reaction of Hwith the conjugate base: Provide Feedback Continue H+ + CH, COO CH, COOH Similarly, any added OH ions will be neutralized by a reaction with the conjugate acid OH + CH3 COOHCHCOO + H2O This buffer system is described by the Henderson-Hasselbalch equation [conjugate base] pH = pK. log [conjugate acid)