CHMB41H3 Lecture Notes - Lecture 28: Ion, Van Der Waals Strain, Markovnikov'S Rule
Document Summary
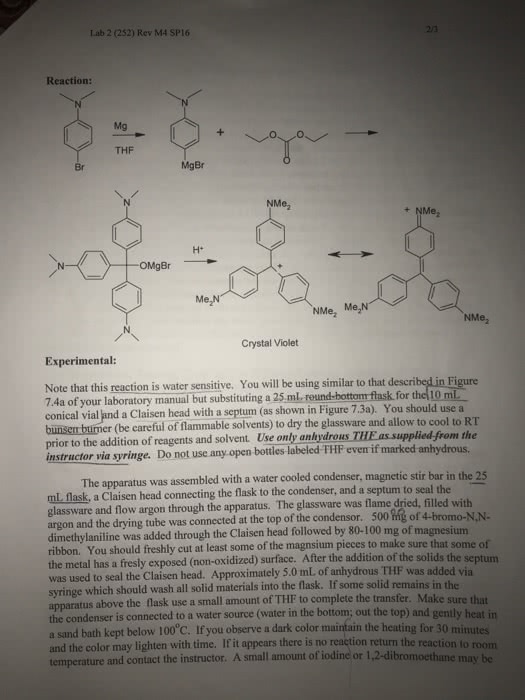
H will add to c with most hs & och3 will add to most substituted carbon. If you add water to alkene you will get alcohol. If you add alcohol to alkene you get ether. Both follow markovnikov"s rule - make a carbon cation. Oxymercuration is more feasible to make alcohol. 1 and 2 = reaction happen in sequence. No strong acids or high temperatures are needed: if your molecule is sensitive to high heat it wont be destroyed because this mechanism allows the reaction to happen without heat. When you have a non symmetrical alkene the o will always attack the most substituted carbon its also anti addition. We end up making alcohol without: carbocation, heat, acids. When # of h increases = reduction. H adds to carbon with most h. O adds to carbon with least number of h. Not using acids = not having carboncations. First line results in a 1,2 methyl shift - results in 3".